Food incident response process
On this page
- Introduction
- 1.0 Conduct a preliminary assessment
- 2.0 Conduct a food safety investigation
- 3.0 Assess the risk
- 4.0 Make a decision
- 5.0 Mitigate the risk
- 5.1 Communicate the risk mitigation plan
- 5.1.1 Notify the lead investigator
- 5.1.2 Communicate the risk mitigation decision to the regulated party
- 5.1.3 Communicate the risk mitigation decision to the inspector responsible for overseeing the regulated party
- 5.1.4 Mandatory recall order
- 5.1.5 Recall of food representing various levels of health risk
- 5.2 Implement the risk mitigation plan
- 5.3 Verify completeness and effectiveness of recall actions
- 5.4 Activities to be conducting following incident response actions
- 5.1 Communicate the risk mitigation plan
- 6.0 Close the incident file
- Appendix 1: Definitions
- Appendix 2: Procedures
- Procedure 1: Recall verification
- Procedure 2: Best practices for storing food safety investigation information
- Procedure 3: Tampering
- Procedure 4: Requesting information from regulated parties during food safety investigations and recalls
- Procedure 5: Documenting key decisions during food safety investigations and recalls
- Procedure 6: Expediting the issuance of a public warning
- Procedure 7: Responding to a company's request for a copy of a risk assessment
- Procedure 8: CFIA guidelines for early public communication of food safety incidents
- Procedure 9: Mandatory recalls
- Procedure 10: Determining after-hours work when responding to food safety incidents
- Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall
- Appendix 3: Templates / Forms
- Template 1: Food safety investigation plan recording form
- Template 2: Letter template for a confirmation of a recall
- Template 3: Letter template for a company initiated recall
- Template 4: Notice of recall
- Template 5: Letter template to distributors requesting recall action
- Template 6: Letter template for a confirmation of a market withdrawal
- Appendix 4: Tables
Introduction
Mitigating risks to food safety is the Canadian Food Inspection Agency (CFIA)'s highest priority. The CFIA works toward protecting Canadians from preventable health risks related to food and zoonotic diseases. The ability to prevent and/or respond to food incidents in a timely and appropriate manner is a critical component to safeguarding food, which enhances the health and well-being of Canada's people, environment and economy.
Purpose
The purpose of this document is to provide guidance to the CFIA inspectorate with respect to the food incident response process. This document will continue to evolve over time. The CFIA will review and update the process, including policies and procedures as required.
Food incidents require CFIA staff to combine the application of rigorous scientific methods and the implementation of sound control procedures, sometimes under the scrutiny of public attention.
Industry is responsible for selling and distributing safe food. It also has key roles and responsibilities including taking appropriate action, such as a food recall, if unsafe food has reached the marketplace. To address this, the CFIA has developed the Recall procedure: a guide for food businesses.
Scope
This process applies to all control actions related to incidents involving food. It is to be used in conjunction with other operational guidance, policies, guidelines and training materials.
References
- Food preventive control and traceability inspection – Compliance verification system
- Food regulatory response guidelines
- Integrated agency inspection model
- Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687)
- Operational procedure: Responding to food complaints (accessible only on the Government of Canada network – RDIMS 13899359)
- Standard incident response process (to be developed)
- Standard inspection process
- Standard regulatory response process
Context
The food incident response process is part of the Standard incident response process.
Control actions, such as a recall, can be taken in conjunction with compliance actions (for example, issuing a letter of non-compliance) and enforcement actions (for example, licence suspension/cancellation or issuance of administrative monetary penalties) during the regulatory response.
Authorities
CFIA inspectors have inspection authorities under the Canadian Food Inspection Agency Act, the Food and Drugs Act and the Safe Food for Canadians Act. These acts provide them with powers to conduct control activities and request control actions.
Roles and responsibilities
Operations Branch
Operations Branch sets operational policies and procedures and provides guidance on how to carry out inspection programs. The branch also verifies industry compliance against requirements; initiates control measures in response to risk; and takes enforcement action in response to non-compliance.
Responsibilities relating to food incident and response, including recalls, are exercised by the Area senior directors and directors of Operations and the director of the Office of Food Safety and Recall (OFSR).
The senior directors and directors of Operations are responsible for ensuring that food safety investigations and recall activities are carried out in a timely, appropriate and effective manner through the application of operational policies and procedures, and that appropriate control measures and enforcement actions are implemented. They are also responsible for ensuring that incidents are referred to the OFSR according to policies and guidelines.
When an incident is referred to the OFSR, the director of OFSR is accountable for its national coordination and for providing consistent advice and direction relating to key aspects of the food safety investigation (such as, determining the scope of affected products, developing sampling plans, etc.) and for making determinations on food recalls and risk communication.
Office of Food Safety and Recall
The OFSR is the national contact point with internal and external partners in respect of food safety investigations and recalls. The office is also Canada's Emergency Contact Point within the International Food Safety Authorities Network (the global network of national food safety authorities managed jointly by the World Health Organization and the Food and Agriculture Organization of the United Nations).
External partners include Health Canada, the Public Health Agency of Canada and international regulatory authorities. Internal partners include other CFIA branches, such as Policy and Programs, Science, Communications and Public Affairs, International Markets and Trade, and Legal Services.
The OFSR is comprised of 3 units, as follows:
A) Food Safety Investigation and Recall Unit
Incidents referred to the OFSR are received by the Food Safety Investigation and Recall Unit, which is responsible for the national coordination of food safety investigation and recall activities. The unit provides advice and guidance to the inspectorate for any aspect of the food safety investigation, and coordinates key steps and decision making relating to the risk response. This includes guiding activities to determine the root cause and the extent of affected products; developing sampling plans; and determining appropriate food recall actions and risk communication. These activities are done with technical support provided by the Food Safety Technical Assessment and Analysis Unit.
The Food Safety Investigation and Recall Unit determines appropriate recall actions based on the level of risk and by applying recall policies and criteria outlined in this guidance and, as appropriate, in consultation with the director of OFSR.
The unit also undertakes specific recall activities, such as the development and posting of public warnings on the Recall and safety alerts website, and liaises with international partners, as required, in respect of food recalls.
B) Food Safety Technical Assessment and Analysis Unit
The Food Safety Technical Assessment and Analysis Unit provides scientific advice to the Food Safety Investigation and Recall Unit and the inspectorate in support of food safety investigations and recall activities. The unit is CFIA's contact point with Health Canada for incident-specific health risk assessments and health risk opinions. The unit reviews and evaluates food safety investigation findings and other risk-related evidence; provides risk estimates for operational requirement; and provides technical expertise in support of key steps and decision making (such as, providing guidance to the Food Safety Investigation and Recall Unit to determine the extent of affected products, developing sampling plans, etc.). When a risk assessment is required for the purpose of making a recall determination, the Food Safety Technical Assessment Unit provides technical risk assessments based on existing Health Canada guidelines, policies, standards or previous health risk assessments or submits requests to Health Canada for health risk assessments.
C) Process Management and Liaison Unit
The Process Management and Liaison Unit is responsible for developing operational policies, procedures and guidance relating to food safety investigation and recalls. The unit develops and reviews material in consultation with the Food Safety Investigation and Recall and the Food Safety Technical Assessment and Analysis Units, the inspectorate and other stakeholders; disseminates and socializes new practices; and develops training material. The unit is also responsible for identifying, maintaining and communicating performance indicators for the CFIA's food safety investigation and recall function. As the administrator of the Issues Management System (IMS), the unit extracts, analyzes and provides food safety investigation and recall data to other branches within the Agency, the media and other external users (for example, through Access to Information and Privacy requests) and identifies trends to inform resource allocation, program policies, foreign verification activities, etc. The unit is the liaison function with other CFIA branches and international partners in respect of food safety investigation and recall-related policies and processes.
Areas
The Areas ensure that food safety investigations adhere to established policies and procedures; effective measures are implemented to mitigate risks posed by food; and appropriate enforcement actions are taken to respond to non-compliance and prevent reoccurrence.
A) Area/regional recall coordinator
The area/regional recall coordinator is the primary contact between area operations staff and the OFSR. They coordinate and monitor food safety investigations at the area/regional level and provide advice and guidance to area/regional staff in respect of food safety investigations and recalls. They assess incident files to determine if a referral is required and subsequently refer those incidents to the OFSR, as necessary. They also brief area senior management, as required; assist in recall implementation; and liaise with provincial government partners.
B) Lead investigator
The lead investigator is responsible for the completeness and accuracy of the food safety investigation and notifying their area/regional recall coordinator of food safety incidents, as required. The lead investigator is the single point of contactFootnote 1 with the regulated party communicating the recommended risk mitigation strategy to the regulated party (such as, requests to conduct voluntary food recalls or serve recall orders). The lead investigator also monitors recall implementation; coordinates verification and follow-up activities; and ensures food safety investigations are expanded to the appropriate extent so that all food that may pose a health risk to the public is investigated and assessed.
The lead investigator may work with other inspectors who assist in food safety investigations and may liaise with other government departments and, when necessary, with police authorities to collect additional information, verify accuracy and completeness of facts or ask for their collaboration for the verification of the effectiveness of recalls.
C) Area Enforcement and Investigative Services (EIS)
EIS will take the lead on facilitating coordination with the appropriate law enforcement agencies in situation where a food safety incident is suspected to be related to criminal activity (for example, tampering or terrorism).
Other branches
A) Science
Science Branch is responsible for providing scientific advice; coordinating testing activities; testing for food and environmental samples, which includes the use of other laboratories; and interpreting and communicating laboratory results of sampling for foodborne illness outbreaks and food safety investigations.
B) Policy and Programs
The Policy and Programs Branch is responsible for providing program direction, advice and guidance and interpreting program requirements. Such advice may be sought from the OFSR in support of complex incidents or from the Food Operational Guidance and Expertise Unit in support of corrective action activities conducted by the inspectorate.
C) International Markets and Trade
The International Markets and Trade Branch is responsible for assisting the OFSR in collecting information related to international food safety investigations and recalls and in notifying countries that are not members of the International Food Safety Authorities Network of food recalls.
D) Communications and Public Affairs
Communications and Public Affairs Branch is responsible for providing risk communication advice; recommending appropriate strategies for dissemination of information; arranging for appropriate media distribution and posting on the CFIA's social media channels, as required; and working with their counterparts at Health Canada, the Public Health Agency of Canada and provincial and territorial governments, as necessary.
E) Legal Services
Legal Services is responsible for providing legal advice, upon request, to CFIA officials during voluntary and mandatory food recalls, including the interpretation and application of legislation; the review of external communications; and representation of the CFIA in communications with third party lawyers.
External partners
A) Health Canada
Health Canada is responsible for setting the regulations and standards for the safety and nutritional quality of food sold in Canada. They provide health risk assessments, as required, to the CFIA and deliver scientific advice and analytical surge capacity. They also develop risk-based guidelines in support of incident-specific risk characterizations.
B) Public Health Agency of Canada
The Public Health Agency of Canada is responsible for the coordination of multi-jurisdictional outbreak response in collaboration with affected partners. They conduct national laboratory-based surveillance; provide technical expertise with respect to outbreak investigations; and interpret and provide feedback on the strength of evidence collected during the epidemiological investigation of a foodborne illness outbreak. The Public Health Agency of Canada acts as the International Health Regulations national focal point and liaises with the OFSR during foodborne illness outbreak investigations. They administer the application of the Foodborne Illness Outbreak Response Protocol.
C) Provincial and territorial government authorities
Provincial and territorial governments have legislation relating to foods produced and sold within their own jurisdictions and are responsible for the associated inspection program at food processors, food service establishments, food retailers, hospitals, long-term care facilities, community kitchens and food banks. They are also responsible for conducting enteric illness surveillance; investigating and controlling human illness outbreaks that occur within their boundaries and for reporting enteric illnesses to provincial/territorial health officials under disease control legislation.
D) Other agencies and organizations
Expertise from other federal, provincial and territorial agencies may be required to provide advice in the control of food safety incidents caused by unusual pathogens or toxic substances in food. Interaction with international partners, such as the International Food Safety Authorities Network, promotes rapid exchange of information with respect to food safety incidents of global interest. Other international partners that work routinely with the CFIA with respect to food safety incidents involving international trade include the European Food Safety Authority, the Pan American Health Organization, the U.S Food and Drug Administration, and the U.S. Centers for Disease Control and Prevention.
If a food safety incident is suspected to be related to criminal activity (for example, tampering or terrorism), law enforcement agencies, such as local police or the Royal Canadian Mounted Police, assume responsibility for the law enforcement response and the criminal investigation.
E) Industry
Industry is responsible for ensuring food safety legislation is followed accordingly so that food sold is safe; assisting the CFIA with the food safety investigation and/or conducting their own food safety investigation; and recalling food effectively from the marketplace. Industry should inform the CFIA when they are removing food from the marketplace. Immediate notification to the CFIA is to be undertaken by companies who are subject to the Safe Food for Canadians Regulations when it is determined that a food presents a risk of injury to human health and if a food is being recalled.
Overview of the food incident response process
The food incident response process, illustrated in Figure 1, encompasses whether a food safety investigation is required, the risk assessment and the development and implementation of risk mitigation measures.
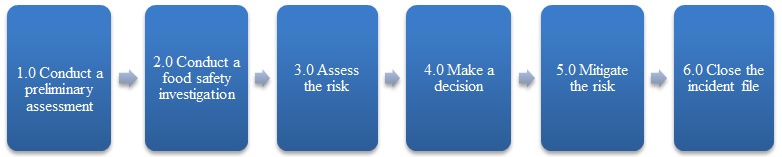
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file.
Although Figure 1 depicts a linear process, for most situations, the steps within the process may be repeated, postponed, revisited or executed simultaneously.
The CFIA's guiding principles related to the food incident response process are as follows:
- Conduct food safety investigations thoroughly, identifying affected or potentially affected food, and use a systematic approach for tracing food (traceback and traceforward)
- Respond appropriately; the depth and breadth of a food incident response will be proportional to the nature of the hazard and likelihood of occurrence
- Conduct food safety investigations consistently and in accordance with policies, procedures, guidelines and standards
- Incorporate the weighting of evidence and the precautionary principle in the assessment of risk and decision making pertaining to risk mitigation
- Take actions to remove risk exposure from the consumer, both immediate exposure (food in distribution) and potential exposure (food not yet in distribution) in a timely manner
- Verify the implementation of corrective actions by manufacturers and/or importers of non-compliant food
- Document information, evidence and decisions taken to support the response, ensuring transparency with partners and stakeholders
- Ensure appropriate engagement with partners, as early as possible, during a food incident response event
- Ensure transparency with the public through timely communication which identifies hazardous situations and/or affected food
- Contribute to the continuous improvement of program design
The food incident response process is triggered by an incident or alleged incident, where a food may pose a risk to human health or contravene legislation, identified through a variety of sources including:
- consumer/members of the public
- regulated parties
- other government departments
- internal branches within the CFIA recording food safety investigation activities and findings
The food incident response process ends when risk mitigation measures, where needed, have been effectively implemented and verified or when it is determined that the food poses no risk or does not contravene legislation. This process will result in a preventive control inspection at one or more of the applicable regulated parties and may also feed into other CFIA processes (for example, enforcement, program management, establishment-based risk assessment model, etc.) depending on the type of food safety incident and findings.
In certain situations, the mobilization of an emergency response team/activation of an Emergency Operations Centre may be required to manage the incident (please refer to the Food Safety Emergency Response Functional Plan (accessible only on the Government of Canada network – RDIMS 3330962)).
Documentation of the food incident
The IMS is the CFIA's system to record information, actions and decisions related to food incidents.
Please refer to the IMS Business Rules (accessible only on the Government of Canada network) and the IMS User Manual (accessible only on the Government of Canada network – RDIMS 1599661) for the procedure on recording food safety investigation activities and findings in the IMS.
The following guidance documents provide additional information on how to document:
- key decisions relating to food safety investigations and recalls, please follow Appendix 2, Procedure 5: Documenting key decisions during food safety investigations and recalls.
- product disposition, please follow Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687)
- follow-up activities, please follow the Food preventive control and traceability inspection – Compliance verification system guidance
1.0 Conduct a preliminary assessment
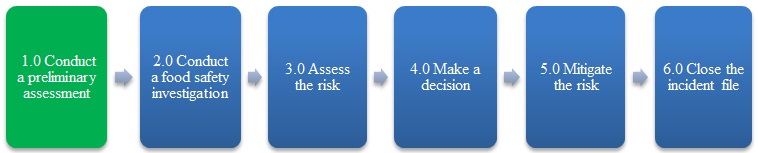
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The first step, conduct a preliminary assessment, is highlighted.
Overview
The purpose of the preliminary assessment is to capture and review the initial information available for an incident involving food in order to: determine whether or not a food safety investigation is required; assign the priority; and identify the next steps for a response.
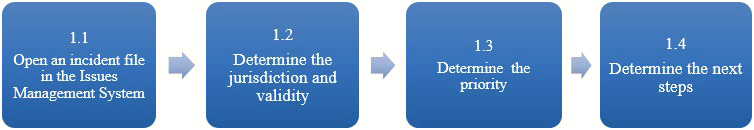
Description of Figure 2: Preliminary assessment
Figure 2 is a flow chart that outlines the 4 sub-steps within the section "conduct a preliminary assessment". The sub-steps are: open an incident file in the Issues Management System; determine the jurisdiction, validity and function; determine the priority; and determine the next steps
1.1 Open an incident file in the Issues Management System
The inspectorFootnote 2 opens an incident file in the IMS when the inspectorate is made aware of an incident that may trigger the food incident response process. Such triggers include, but are not limited to:
- Complaints from consumers, industry, associations or referred by other government departments
- Company initiated product action, such as removal of the food from sale or the correction of its label
- Suspected or confirmed foodborne illness outbreakFootnote 3 situations
- Unsatisfactory or investigative laboratory results
- CFIA inspection findings
- Referrals from other branches within the CFIA
- Referrals from other federal/provincial/territorial, municipal and international government departments or food safety organizations (for example, the International Food Safety Authorities Network), law enforcement, the medical community
- Traditional or social media
The Office of Food Safety and Recall (OFSR) will open the incident file in the IMS when the trigger is a referral from an international authority.
If the trigger is a complaint, please follow this manual in conjunction with the Operational procedure: Responding to food complaints (accessible only on the Government of Canada network – RDIMS 13899359).
Depending on the initial trigger, food safety investigations can occur at various levels of the distribution chain, such as consumer, retailer, distributor, manufacturer, importer, etc.
1.2 Determine the jurisdiction and validity
1.2.1 Jurisdiction
The lead investigator will determine whether the incident falls under the responsibility of the CFIA or under an authority outside of the Agency. Depending on the nature of the incident, responsibility may be shared with other federal/provincial/territorial governments.
Once the jurisdiction has been identified, if it falls under the CFIA, the lead investigator is responsible for investigating the regulated parties that are located in their region. Where the incident extends to a regulated party located in another region, the lead investigator will refer it through the IMS to the appropriate region.
The nature of the food involved in the incident will help the lead investigator determine the appropriate jurisdiction, as follows:
Food under the jurisdiction of the CFIA
Food regulated under the Food and Drugs Act and other legislation enforced by the CFIA is considered to fall under the CFIA's jurisdiction. AdviceFootnote 4 can be sought to determine if the affected food is under the jurisdiction of the CFIA.
If it is determined that the incident falls under CFIA jurisdiction, the lead investigator will determine whether it requires the initiation of a food safety investigation.
Shared jurisdiction
For incidents involving shared jurisdiction, the food falls under the CFIA's jurisdiction, but some activities may be conducted by another authority at the local, provincial/territorial or federal level. Examples of such incidents include:
- Food safety investigations in conjunction with provincial, municipal or local health authorities (for example, local public health units, « le ministère de l'Agriculture, des Pêcheries et de l'Alimentation du Québec », Ontario Ministry of Agriculture, Food and Rural Affairs, British Columbia Centre for Disease Control, etc.). In some situations, the shared jurisdiction may be defined by a Memorandum of Understanding.
- Incidents involving the United States (United States Food and Drug Administration or the United States Department of Agriculture), as required.
-
Foodborne illness cluster or outbreak investigations where the suspect food is under the CFIA's jurisdiction. The food safety investigation will be conducted by the CFIA; however, the illness and epidemiological investigation will be conducted by public health partners involved in the outbreak investigation.
For more information, please refer to the foodborne illness outbreak response protocol.
-
Food tampering incidents may include a food safety investigation as well as a police investigation. The CFIA plays an important role in the initial identification of tampering incidents and in their ultimate resolution from a food safety perspective, while the primary responsibility for the investigation of the perpetrator rests with police authorities.
For more information and procedures on tampering, please follow Appendix 2, Procedure 3: Tampering.
-
Food prepared in restaurants, institutions or retail stores is initially investigated by the appropriate public health authority and may subsequently be referred to the CFIA depending upon their findings.
Food not under the jurisdiction of the CFIA
The following items do not fall under the CFIA's jurisdiction:
- Foods marketed as natural health products
- Food served on passenger conveyances (for example, airlines, passenger rail and marine)
- Food served in federal facilities (for example, prisons)
- Municipal and recreational water
The inspectorate refers all available information to the appropriate authority (for example, Health Canada, the local public health unit, etc.). Once the file has been referred, the lead investigator proceeds to Section 6.0 – Close the incident file in the Issues Management System.
1.2.2 Validity
Using the preliminary information available, the lead investigator will confirm the validity of the incident by determining if the food has the potential to represent a health risk and/or contravenes applicable legislation.
The lead investigator will use judgement, training and experience when determining the validity of the incident and will seek adviceFootnote 5, as necessary.
Determining the validity of an incident can be ongoing during the food safety investigation and as more information is obtained, the incident may be found to not be a valid concern.
If the incident is not a valid concern (for example, a consumer complaining about the shape of a cookie), the lead investigator proceeds to Section 6.0 – Close the incident file in the Issues Management System.
If the incident is valid, but does not require a food safety investigation, the lead investigator will follow the appropriate guidance documents for compliance verification, enforcement and/or food labelling.
If it is not clear if the incident is valid, the lead investigator will conduct the food safety investigation, as described in this document, until it is confirmed that the food does not represent a health risk, contravenes legislation or until the process is completed.
1.3 Determine the priority
Prioritization is an important aspect of the food incident response process as it determines the urgency with which the CFIA will respond to an incident.
The priority is assigned by the lead investigator based on available information and the following factors:
- The hazard (for example, microbial, chemical, allergen, extraneous material)
- Hazard level (the severity, likelihood of harm to consumers)
- Distribution (is the food on the market?)
- Target population (for example, infants, toddlers, elderly, immunocompromised)
- Reported illness, injury or reaction
- Shelf life of the food
For food complaints, follow the food complaint procedure (accessible only on the Government of Canada network – RDIMS 13899359) to determine the appropriate priority and pathway.
If further expertise is required to determine the priority, the lead investigator will consult with their supervisor and the area/regional recall coordinator, who may in turn consult with the OFSR.
The priority of food safety incidents is defined as follows:
- High priority: A food safety incident that takes precedence over all other work by Operations Branch.
- Normal priority: A food safety incident that is not a high priority.
For a complete list of high priority food safety incidents, please follow Appendix 2, Procedure 11, Annex A: High priority food safety incidents with requirements for early referral.
It is important to note that the priority assigned to an incident can change due to new findings during the food safety investigation (in other words, a normal priority incident can become a high priority incident and vice versa).
1.4 Determine the next steps
The lead investigator will report all potential food safety incidents, including low risk incidents such as non-harmful extraneous material, extensive insect contamination, spoilage and other similar incidents, to their supervisor. Potential or confirmed high priority food safety incidents will be reported immediately to the area/regional recall coordinator.
When a food does not represent a health risk, but there is a non-compliance, the lead investigator will take appropriate compliance actions according to the Standard inspection process and/or other appropriate guidance.
1.4.1 Referring food safety incidents to the Office of Food Safety and Recall
Notifying the area/regional recall coordinator who in turn refers food safety incidents to the OFSR at the appropriate time ensures an efficient and consistent CFIA response to potential risks of injury to human health associated with food in distribution. It enables:
- engagement of appropriate team members (for example, the lead investigator, supervisor, area/regional recall coordinator, OFSR) at the right time
- communication of appropriate information in a timely manner within the food safety investigation team with CFIA's senior management, other CFIA branches and external partners (for example, Health Canada, the Public Health Agency of Canada, the U.S. Food and Drug Administration, the United States Department of Agriculture and the International Food Safety Authorities NetworkFootnote 6)
- a more effective use of operational resources by limiting delays and redundancies.
There are situations where an incident is brought to the attention of the OFSR first, either by CFIA senior management, other CFIA branches, or external partners (such as the Public Health Agency of Canada, foreign governments or international organizations). In such cases, the OFSR engages the inspectorate, without delay, by communicating the information to the area/regional recall coordinator.
For the process for referring food safety incidents, including early referrals, to the Office
of Food Safety and Recall, please follow Appendix 2, Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall.
1.4.2 Immediate control actions
The lead investigator, in consultation with their supervisor, inspection manager, area/regional recall coordinator, etc., will determine whether certain immediate control actions, such as seizure and detention, are required to mitigate the potential health risk. Such control actions can be initiated at any stage of the food incident response process and may need to be implemented before a risk mitigation plan is developed. The regulated party can also take immediate control actions, such as product disposition.
If the situation requires an immediate enforcement action, the lead investigator should follow section 5.2 of the Standard regulatory response process and the Enforcement referral and triage standard, as appropriate.
Certain high priority food safety incidents may require public communication in the early stages of the food safety investigation (in other words, before the level of health risk has been established and/or a food recall is implemented). The OFSR, in collaboration with Communications and Public Affairs Branch, will make the decision following Appendix 2, Procedure 8: CFIA guidelines for early public communication of food safety incidents, which outlines the 2 types of situations where early communication may be considered and the procedure to follow:
- Early public communication to respond to food tampering situations
- Early public communication to address concerns raised by the public regarding food safety incidents.
In such cases, the lead investigator will ensure that the regulated party is advised of the public communication.
1.4.3 After-hours work
It is important for management responsible for Operations Branch resources to effectively respond to food safety incidents and allocate resources in a way to achieve food safety objectives. There are a number of stakeholders engaged within Operations Branch to address food safety incidents. Strong collaboration is important between these units in order to provide support as required and ensure a seamless and efficient food safety response.
At all times, high priority food safety incidents will take precedence over other work during core business hours with a view to minimize possible after-hours work. In certain situations, working outside of core business hours may be required by the area(s), by the OFSR or by both.
For guidance on determining which food safety incidents are to be addressed outside of core business hours including factors to be considered in making the decision, please follow Appendix 2, Procedure 10: Determining after-hours work when responding to food safety incidents.
2.0 Conduct a food safety investigation
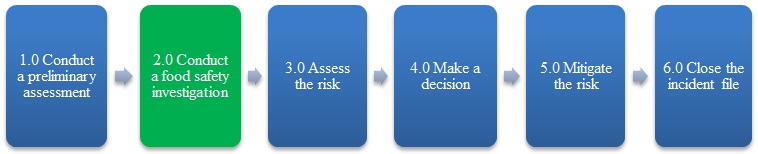
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The second step, conduct a food safety investigation, is highlighted.
Overview
A food safety investigationFootnote 6 is undertaken by regulatory officials to verify whether a contravention or a hazard, which could cause human injury or illness, exists in a food and to determine the nature and extent of the problem. Information collected during a food safety investigation provides the basis for risk assessment.
It is not a regulatory investigation which is conducted in response to non-compliance in order to recommend further compliance measures or enforcement action.
The food safety investigation will continue until complete and sufficient information is gathered to develop an appropriate risk mitigation strategy. In some situations, the food safety investigation may determine that the food does not pose a risk, however, further action may be required. The food safety investigation may continue as some risk mitigation actions are implemented, such as a food recall, to address all aspects of the incident and the full scope of affected food.
During the food safety investigation, if it is determined that the incident does not represent a potential health hazard, the lead investigator will:
- inform the inspector responsible for overseeing the regulated party who will follow the appropriate guidance
- refer to Section 6.0: Close the incident file in the Issues Management System if no potential health risk nor violation has been identified

Description of Figure 3: Food safety investigation
Figure 3 is a flow chart that outlines the 5 sub-steps within the section "conduct a food safety investigation". The sub-steps are: develop a food safety investigation plan; prepare for the food safety investigation; collect information; verify and analyze data/information; and scope the incident.
The key principles of food safety investigations are:
- Timeliness: Food safety incidents are investigated promptly with time frames proportional to the potential risk while taking the shelf life of the food into consideration.
- Appropriateness: The depth of food safety investigations is appropriate to the hazard and the likelihood of its occurrence. Food safety investigations cover all relevant levels at which the food has been manufactured, imported, distributed, sold or consumed.
- Consistency: Food safety investigation procedures are consistent with internal policies, procedures, guidelines and standards and based on scientific rigour.
- Thoroughness: Food safety investigations identify all affected or potentially affected food. A systematic approach is utilized for tracing food forward and backward within the distribution system.
- Accuracy: Food safety investigations strive to detect and identify the hazard in all affected or potentially affected food that may pose a food safety risk to the Canadian public. Information collected should facilitate and inform decision making by being as accurate and concise as possible.
- Partnership: Collaboration and cooperation among stakeholders is necessary to ensure food safety investigation activities are conducted in a timely, effective and appropriate manner.
- Transparency: Disclosure of information pertinent to the food safety investigation, while adhering to access to information and privacy laws, is needed to ensure all stakeholders are able to fulfill their responsibilities to the best of their abilities.
2.1 Develop a food safety investigation plan
Planning is a key element in all food safety investigations, irrespective of the trigger. After it has been determined that a food safety investigation is required, the lead investigator or food safety investigation team, as appropriate, will start by preparing a food safety investigation plan. The plan will help enhance the coordination, efficiency and timeliness of the food safety investigation as well as clarify its goals.
2.1.1 Focus on the area of concern
For each food safety investigation, it is important to focus on the area(s) of concern related to the incident in order to collect relevant information. Food safety incidents fall under one or more of the following main areas of concern:
- microbiological
- allergens
- chemical
- extraneous material
- nutrition
Examples of questions to ask during the food safety investigation related to these areas of concern and a core information check list are found in the Food Safety Investigation Job Aids Folder.
2.1.2 Conduct a fact finding check
The lead investigator will conduct a fact finding check with regard to the food, the regulated party and the process.
-
Food
The lead investigator will verify the applicable legislation, guidelines and policies as well as identify the common concerns associated with the food or type of food.
-
Regulated party
The lead investigator will review the history of the regulated party involved together with the nature of their operations.
-
Process
The lead investigator will review and familiarize themselves with the processes used to manufacture, if applicable, and handle the food.
The lead investigator should also search in databases such as the Issues Management System (IMS), the Import Retrieval System, the Digital Service Delivery Platform (DSDP) and the Laboratory Sample Tracking System for historical information related to the food of interest and the regulated party responsible for its manufacture or importation.
2.1.3 Consultation
The lead investigator will consult with their supervisor, area/regional recall coordinator, the Office of Food Safety and Recall (OFSR), other inspectors and/or operational specialists as appropriate. The food safety incident may have similarities with previous incidents and their expertise and guidance may help inform the food safety investigation plan (for example, line of inquiries or questions, precedents or history, etc.).
2.1.4 Food safety investigation team
The lead investigator and/or the area/regional recall coordinator, with the support of their respective supervisors and managers, will determine if a food safety investigation team is necessaryFootnote 7 based on their preliminary assessment of the potential scale of the incident. The following will be taken into consideration:
- Risk category of the food
- Nature and complexity of the incident
- If there are associated/suspected illnesses
- Volume of production
- Quantity and type of food in distribution
- Extent of distribution (local, national or international)
- Compliance history (risk rating) of the regulated party
- Size of the regulated party's facility
- Regulated party's available resources to support the food safety investigation
As the food safety investigation progresses, the lead investigator will consider if there are other individuals who should be involved to provide expertise or assistance and whether they need to be integrated into the food safety investigation team.
Depending on the scale of the investigation and the volume of information to be collected, consideration should be given to identifying team member(s) to assist with data collation and analysis.
The food safety investigation team can consist solely of the lead investigator or may include regional, such as other inspectors, area and headquarters staff (for example, the OFSR). Each team member will actively contribute to the food safety investigation by undertaking specific tasks. Members may be engaged or disengaged as required.
2.1.5 Draft the food safety investigation plan
After it has been determined that a food safety investigation is warranted, the lead investigator, in consultation with the food safety investigation team, when appropriate, will prepare a food safety investigation plan. A food safety investigation plan is a strategy on how to approach and conduct the food safety investigation. The plan will specify the areas to be investigated and the activities to be conducted.
The plan should include, as appropriate:
- Food safety investigation objectives that summarize the broad goals to be achieved
- A list of key questions relevant to the area of concern
- Key areas to be investigated, such as:
- Equipment and structures
- Possible internal and external sources and routes of contamination.
- Type of investigational activities required for each key area, such as:
- Food and environmental verification
- On-site inspection activities
- The assistance of a local public health unit (for example, a facility in a remote location)
- Traceback/traceforward
- A list of equipment that may be needed during the food safety investigation, such as:
- regular inspection equipment (for example, personal protective equipment)
- specialized equipment
- a camera or smartphone for taking clear photos of the food label
- Relevant documents such as appropriate legislation
- Any special considerations, such as legal considerations, and expertise required
- An outline of communication activities (for example, request for information from a regulated party, reports/briefings to management)
- Timelines for the food safety investigation and for each key area to be investigated, including:
- a start date which depends on the priority of the food safety incident and availability of CFIA staff and whether after-hours work is required (refer to Appendix 2, Procedure 10: Determining after-hours work when responding to food safety incidents)
- an anticipated completion date which is based on a reasonable and objective assessment of the time required to document the results of each area to be investigated.
A documented plan is preferable (refer to Appendix 3, Template 1: Food safety investigation plan recording form); however, a verbal discussion with the supervisor outlining the next steps may be sufficient.
As the food safety investigation progresses, the lead investigator will modify the food safety investigation plan as necessary and depending on the information that is collected.
2.1.6 Validation of the food safety investigation plan
The lead investigator will present the plan to his or her supervisor for review and to verify that there are no missing lines of inquiry which may cause gaps in the information collected. The area/regional recall coordinator will be consulted as necessary.
2.2 Prepare for the food safety investigation
2.2.1 Assemble the food safety investigation team
The lead investigator, in consultation with his or her supervisor, selects additional team members, as needed, based on skill sets, training and experience to assist in the food safety investigation.
For high priority food safety incidents, the lead investigator will engage the area/regional recall coordinator and the OFSR as team members as per Appendix 2, Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall.
2.2.2 Gather the equipment and tools required
The lead investigator will prepare and assemble the required equipment, tools and documents for the food safety investigation.
2.3 Collect information
The information collected during a food safety investigation, is used to determine the potential health risk posed by the food, identify if other foods are affected and verify that the root cause has been identified.
When an incident file has been referred to the OFSR, they will coordinate food safety investigation activities to ensure information collected is thorough, complete and accurate to inform risk mitigation decisions. When an incident file has not been referred to the OFSR, the lead investigator will coordinate the food safety investigation activities.
The lead investigator or assigned inspector will:
- collect information on-site when possible and according to the food safety investigation plan. Information should be gathered by observation, interviews and examination of records and documents
- record detailed, accurate and complete notes and organize them in a manner that facilitates root cause verification, data/information analysis, risk assessment, decision making and the development of risk mitigation measures.
For information on:
- notifying the complainant, follow the Operational procedure: Responding to food complaints (accessible only on the Government of Canada network – RDIMS 13899359)
- requesting information from the regulated party, follow Appendix 2, Procedure 4: Requesting information from regulated parties during food safety investigations and recalls
- documenting in the IMS, follow the Business rules of use for the IMS
For all food safety investigations, the lead investigator or assigned inspector will collect the following core information:
- Food: name, format, storage condition (for example, shelf-stable, refrigerated, frozen), shelf life/durable date, codes, packaging, copy of the food label, country of origin and foreign manufacturer information, where appropriate
- Regulated Party: type of company, contact details, return policy
- Production: quantities, codes, processing, packaging, controls
- Distribution: quantities, geographical distribution, including whether the food has been exported, type of receiving clients, if the food was sold to a cannabis licence holder
- Complaint records from the regulated party
- Any reported reaction/injury/illness
The lead investigator will also collect specific information related to the area of concern.
Please refer to Appendix 2, Procedure 2 for best practices for storing food safety investigation information.
2.3.1 Follow the suspect food
A food safety investigation can be conducted by "following the suspect food" at any point in the distribution chain (consumer, retailer, distributor, hotel/restaurant/institution, manufacturer and importer levels). The lead investigator will conduct traceback and traceforward activities as necessary to ensure complete and accurate verification of information (affected food, root cause, etc.) associated with the food safety incident. Before proceeding to the next point in the distribution chain, the lead investigator will ensure that the current distribution level where the investigation is being conducted is not the source of the problem.
When the food safety investigation is being conducted at the point of manufacture, the process of production should be followed from start to finish, as applicable. In other circumstances in the manufacturing plant where cross-contamination may occur, the food should be investigated from the finished state back to the source of input.
2.3.2 Root cause analysis
The root cause is the original source of or reason for the incident. Root cause analysis consists of determining where, when and how the incident occurred. Identifying the root cause will allow the development of appropriate corrective measures to prevent the recurrence of the deviation. For the purpose of the food safety investigation, identifying the root cause will help determine the scope of the incident (in other words, attribute a risk to other food based on food safety investigation findings).
The root cause may be deviations in the process, environment, equipment, personnel and incoming materials.
The lead investigator will verify that the regulated party has "followed the suspect food", as described in Section 2.3.1 or via any other appropriate means, to identify where the deviation occurred and at each step will answer the following:
- Is there a possibility that the deviation occurred at this step?
- Were there control measures to prevent the introduction/control of the hazard? If so, were they sufficient and implemented adequately?
In situations where the regulated party has already determined the root cause, the lead investigator will initiate a document review to ensure the root cause was appropriately identified.
Additionally, the lead investigator will determine if the root cause is attributed to any of the following:
- Employee practices
- Improper sanitation or maintenance
- Inadequate cross contamination controls (process flow, plant floor layout, etc.)
- Improper processing and/or
- Incoming materials
If the root cause has not been identified during the food safety investigation, it may be found as part of the activities conducted following incident response. Refer to the Food preventive control and traceability inspection – Compliance verification system guidance.
2.3.3 Sampling
Laboratory analysis of environmental and food samples can be an important source of information during food safety investigations. It can be used to verify the existence of a hazard and/or to determine the extent of affected food. Sampling may provide evidence to confirm or identify the root cause of an incident, assist in the risk assessment and identify the focus of the food safety investigation. Sampling can be conducted at any time during a food safety investigation (from the trigger onwards).
Sampling builds on document verification (for example, record review), direct observations and inspection findings related to the suspect food. Document verification helps to determine if the suspect food was made under normal conditions; to understand any limitations; and to identify any incidents that show points of possible contamination.
The lead investigator, in consultation with the OFSR or the food safety investigation team, as appropriate, should consider if sampling is necessary. It may be necessary to communicate with the appropriate laboratory or with the Laboratory Coordination Division for additional instructions and availability of laboratory testing, as per area/regional protocols.
Investigational sampling of food and the environment aims to identify all affected food and potential sources of contamination, such as a food contact surface. This is done by sampling as close to the point of possible contamination and may be extended to determine the scope of the incident. The number of samples to be analyzed must be sufficient to reach a certain level of confidence in the detection and/or quantification of the hazard. Investigational samples may not necessarily represent the potentially implicated lot in statistical terms; however, they would determine if an entire lot is acceptable or unacceptable for human consumption. It is important that the manufacturer or distributor provides key information, such as the quantity of units produced, distribution and availability of the food to allow the appropriate selection of investigational samples.
Food test results from industry sources should be considered. The CFIA must be satisfied that the samples represent the lot (in other words, the results allow CFIA to determine if the entire lot is acceptable or unacceptable) and that the sampling methods, testing methods and laboratories used are accredited and equivalent to the Canadian standards.
To optimize sampling, the lead investigator may consult with their supervisor, the area/regional recall coordinator, the area operational specialist, the Laboratory Coordination Division, etc., on methodology and in selecting the best sample site and size in order to enhance the determination of whether a hazard is present in the food. When a food safety incident has been referred to the OFSR, the lead investigator will consult with the OFSR on required sampling. The OFSR provides sampling plans based on, but not limited to; document verification, regulated party history, the nature of the hazard, internal and external standards, peer reviewed scientific literature and hazard/commodity history and will communicate with the Laboratory Coordination Division.
In situations where multiple regions in one area are involved in sampling activities, the area will identify a sampling coordinator. In other situations, when multiple areas or regulated parties are involved; the OFSR can coordinate sampling activities.
For more information on sampling, please refer to the CFIA's sampling and testing policy.
2.3.4 Information sharing
Information collected during the food safety investigation can be exchanged between various internal and external partners according to Memorandums of Understanding and existing procedures. For more details on the type of information that can be shared by the CFIA with other government departments, regulated parties, third parties, and the public during food safety investigations and recalls, please refer to the CFIA's protocol on information sharing during food safety investigations and recalls.
2.4 Verify and analyze data/information
The lead investigator will analyze the information collected to determine if it is relevant to the food safety incident and the hazard of concern to identify any outstanding gaps and to verify that the information provided verbally or via e-mail by the regulated party can be supported by monitoring records or invoices.
2.5 Scope the incident
The determination of the scope of affected products during a food safety investigation is a set of activities that consists of verifying if there is any other food under control or in distribution that contains or may contain the same or similar hazard as the food initially investigated or recalled.
These activities are not intended to address the safety of the establishment's future production, but are intended to focus on food already produced at the time of the food safety investigation.
This determination is a key element of the CFIA's decision making process and the subsequent risk mitigation measures when responding to a food safety incident.
For incidents involving the presence of E. coli O157:H7 in raw beef, the lead investigator will follow the Operational procedure: Control response plan for the detection of E. coli in raw beef products.
There are 3 main activities undertaken to determine the scope of affected products during a food safety investigation:
- Determine if the root cause has been identified, reviewing the regulated party's investigation findings, as applicable
- Determine the timeframe of interest
- Identify all affected food
2.5.1. Root cause analysis
The lead investigator will review and validate the accuracy of the regulated party's root cause analysis, where undertaken, to assist in determining if and what other food may be affected. If the root cause of the incident was not determined during the food safety investigation or the regulated party did not conduct a root cause analysis, the root cause may be identified as part of the activities to be conducted following incident response actions (please refer to section 5.4 for additional information).
2.5.2. Determine the timeframe of interest
The timeframe is calculated from the date the deviation was corrected or the date the scope starts (end date) backward to the date or the most probable date that the deviation which caused the incident occurred or started (the onset).
Example: The manufacturer started to use contaminated flour on September 25th and stopped using it on October 5th. The timeframe to scope will be from October 5th (end date) backward to September 25th (onset date). All food manufactured during this period of time that used the contaminated flour or that could have been contaminated by the flour are considered potentially affected.
Table 1: General guidelines for the determination of the timeframe to scope provides general guidance on the determination of the timeframe for specific root causes. Depending on the incident, different timeframes can be used and the timeframe can be modified as the food safety investigation progresses. The lead investigator will consult with the area/regional recall coordinators and the OFSR.
In situations where the root cause or the most probable root cause is not possible to identify (for example, regulated party and/or CFIA reports of analysis are satisfactory, no deviations observed, no system failure identified, etc.), the maximum timeframe to use to determine the scope is the date the scope starts backward to the longest shelf life of the affected food.
In situations of extended shelf life of the food, the lead investigator will determine the timeframe by taking the following into account:
- the food safety investigation findings
- results of laboratory analysis (period covered by the sampling)
- records of production
- formulations, when applicable
- history of conformity
- the rapidity of sale of a food
- epidemiological data (sampling of food outside the incubation period should not occur)
2.5.3 Identify all affected food
The set of activities below may take place simultaneously or in a different order. The lead investigator may determine that certain verification activities are not required due to the type of incident and the root cause or the most probable root cause identified.
The lead investigator will consult the area/regional recall coordinator to determine the best approach to identify all affected food.
2.5.3.1 Same product verification
The lead investigator will conduct this verification in all food safety investigations in order to verify if the food initially investigated or recalled is packaged and/or labelled with other codes, other brands or in other container sizes at the establishment where it was produced.
For an imported food, the lead investigator will verify if the food initially investigated or recalled may have been imported under other codes, other brands, and/or packaged in other container sizes.
If the lead investigator determines that the food initially investigated or recalled was packaged and/or imported and/or labelled with other codes, other brands, and/or packaged in other container sizes, these foods are considered affected. The lead investigator will follow the steps described in Section 1.4.1 Referring food safety incidents to the Office of Food Safety and Recall, if the file has not already been referred.
The lead investigator will stop same product verification activities when:
- appropriate control measures have been taken for all affected food identified or
- when they confirm that the food initially investigated or recalled (whether imported or not) was not packaged and/or labelled under other codes, other brands, and/or packaged in other container sizes
2.5.3.2 Other products verification
The lead investigator will conduct this verification when the root cause or the most probable root cause originates from a process, equipment or employee practice that is common with other food produced/packaged/stored at the establishment.
The lead investigator will use the pre-established timeframe to verify if other food manufactured in the establishment or other food imported from the same supplier could have been in contact directly or indirectly with the food initially investigated or recalled.
The lead investigator will examine:
- the flow of food through the facility
- the handling practices at each step
- the conditions in the areas of production
- all related records to identify any discrepancies
When the food is imported, the ability to perform this step may be limited but can be performed via the information provided by the importer or by foreign governments.
If other food is affected, the lead investigator will consult the area/regional recall coordinator or the OFSR (if the file has already been referred) who will provide advice on whether the other affected food should be sent for a health risk assessment (refer to section 3) or should be sampled to confirm the hazard (refer to section 2.3.3).
The lead investigator will stop other products verification activities when:
- appropriate control measures have been taken for all affected food or
- they confirm that there is no other food that could be affected
2.5.3.3 Other processes verification
The lead investigator will conduct this verification when the root cause or the most probable root cause could have affected other processes in the establishment.
The lead investigator will use the pre-established timeframe to verify if the food manufactured under other processes contains or could contain the same hazard as the food initially investigated or recalled.
When the food is imported, the ability to perform this step may be limited but can be performed via the information provided by the importer or by foreign governments.
If the lead investigator identifies other affected food, they will consult the area/regional recall coordinator or the Office of Food Safety (if the file has already been referred) who will provide advice on whether the other affected food should be sent for a health risk assessment (refer to section 3) or should be sampled to confirm the hazard (refer to section 2.3.2).
The lead investigator will stop other process verification activities when:
- control measures have been taken for all affected food
- they confirm that the root cause or most probable root-cause does not impact on other processes and/or
- they confirm that there is no other food that could be affected
2.5.3.4 Other regulated parties verification
Other regulated parties may be affected when:
- the affected food may have been further processed or repackaged
- the raw material is the root cause, as other regulated parties may use the same material
- the food initially investigated or recalled was imported, as other importers may have imported the same food or other affected food
The lead investigator will initiate a new food safety investigation for each new regulated party identified and verify if they have sold food that represents a health risk. The lead investigator may have to refer incidents to inspectors in other regions/areas depending on where the regulated party is located.
The lead investigator and assigned inspectors will stop other regulated party verification activities when:
- all regulated parties identified have been investigated
- all affected food has been assessed and control measures have been taken or
- they confirm that there is no other regulated party that could have been affected by the problem
2.5.4 Communicate and refer the incident to the Office of Food Safety and Recall
If the file has not already been referred, the lead investigator will communicate the incident to the area/regional recall coordinator, as per the area/regional protocol. The area/regional recall coordinator will follow the existing guidance for referring the file to the OFSR as described in Appendix 2, Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall.
| Root cause/most probable root cause | End date instructions (deviation corrected) | Onset date instructions (date deviation started) |
|---|---|---|
| Incoming raw material | The date the deviation was corrected or the regulated party stopped using the raw material | The date the raw material was first used |
| Environment and/or equipment including cross contamination | The date the new production started to be under "Hold and Test" regime or the date the problem was corrected (robust sanitation, equipment corrected etc.) whichever comes first | The date the deviation started or the date calculated from the end date minus the shelf life of the food or the date the last verification was done, whichever comes first |
| Process breakdown (insufficient cooking, cooling, sealing, etc.) | The date the problem was corrected | The date the failure occurred or the date calculated from the end date minus the shelf life of the food, whichever comes first |
| Packaging material | The date the problem was corrected | The date the packaging material started to be used or the date calculated from the end date minus the shelf life of the food, whichever comes first |
| Wrong label or formulation | The date the problem was corrected | The date the wrong formulation or label started to be used or the date calculated from the end date minus the shelf life of the food, whichever comes first |
3.0 Assess the risk
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The third step, assess the risk, is highlighted.
Overview
The findings of the food safety investigation may indicate that the incident represents a potential health risk which needs to be assessed for the purpose of making a risk mitigation decision.
Assessing the risk will guide whether risk mitigation actions are required and which ones would be the most appropriate. When it is known that a food recall will need to be considered as one of the risk mitigation measures, assessing the risk associated with the food in question becomes an essential step. Other risk assessment activities, such as estimating risk, may be required to inform the priority to assign to an incident for response by Operations Branch.
The legislative power in support of food recalls is based on risk as outlined in section 19(1) of the Canadian Food Inspection Agency Act. This Act gives authority to the Minister of Health to mandate a recall where the Minister believes on reasonable grounds that a regulated food poses a risk to public, animal or plant health.
The Canadian Food Inspection Agency (CFIA) and Health Canada have shared responsibilities in food safety. According to a Memorandum of UnderstandingFootnote 8, Health Canada will provide health risk assessments upon request from the CFIA and other organizations. Also according to the Memorandum of Understanding, the CFIA may assess risk associated with food for which Health Canada guidelines, policies and standards are in place and no policy interpretation is required. The Office of Food Safety and Recall (OFSR) is the single contact point with Health Canada for obtaining health risk assessments for the purpose of making recall determinations.
The following figure describes the four main activities involved in the mentioned process.

Description of Figure 4: Assess the risk
Figure 4 is a flow chart that describes the four main activities involved in the section "assess the risk". The activities are: evaluate the need for risk assessment; request a risk assessment; conduct a risk assessment; and receive the risk assessment.
3.1 Evaluate the need for a risk assessment
3.1.1 Triaging by the Food Safety Investigation and Recall Unit
There are a number of situations where risk assessment expertise may be required during a food safety investigation. The Food Safety Investigation and Recall Unit is the entry point into the OFSR via Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall. The food safety recall specialist assigned to the file reviews the incident and the available risk evidence as the food safety investigation progresses, and may seek risk assessment expertise from the Food Safety Technical Assessment and Analysis Unit in the OFSR at different stages in the process and for various purposes. The type of expertise provided by the Food Safety Technical Assessment and Analysis Unit will depend on the situation and the intended use. The situations for which the food safety investigation and recall Unit may need risk assessment expertise are described as follows:
a) To assist with assigning priority to an incident
The food safety recall specialist may seek a risk estimate from the Food Safety Technical Assessment and Analysis Unit to assist with assigning priority to an incident as well as informing decisions on whether or not certain activities associated with a high priority incident should continue outside of core business hours.
b) When evidence collected on an incident suggests that a food recall may be required
When information collected in relation to an incident indicates that a hazard that potentially poses a risk to consumers is present in a food and there is on-going exposure for consumers, the food safety recall specialist will review all information and evidence collected to verify that:
- the presence of a hazard in the food has been confirmed; and
- ongoing exposure for consumers has been established; and
- information is complete enough to conduct a risk assessment
Based on the review, if the food safety recall specialist believes that the situation represents a potential health risk which needs to be assessed for the purpose of making a recall determination, a request for a risk assessment will be made to the Food Safety Technical Assessment and Analysis Unit of the OFSR. The food safety recall specialist may need to seek additional information or clarification from the lead investigator to complete the request.
Note: In special circumstances, the OFSR may decide to proceed to section 3.2: Requesting a health risk assessment, using limited risk evidence information, when a risk mitigation decision is required urgently. Although the health risk assessment may not lead to a conclusive risk classification, elements from the health risk assessment may inform possible precautionary risk mitigating measures with the support of health portfolio partners.
c) When a regulated party has initiated a food recall or other product action
When a regulated party has informed the CFIA that it has initiated action on a food at its own volition with or without a public warning, the food safety recall specialist and the technical risk assessor will review the information relating to the incident and determine whether the product action taken by the regulated party is sufficient for the type of risk and adequately mitigates the risk to consumers. If necessary, the food safety recall specialist will request a risk assessment from the Food Safety Technical Assessment and Analysis Unit.
3.1.2 Triaging by the Food Safety Technical Assessment and Analysis Unit
Upon receipt of a risk assessment request, the assigned technical risk assessor will review the risk evidence available for the incident and relevant risk information (risk based guidelines, previous health risk assessments addressing similar situations, scientific literature, etc.) to provide a response to the Food Safety Investigation and Recall Unit.
For this review, the technical risk assessor may seek additional information or clarification from the food safety recall specialist, as required, who in turn may need to seek information or clarification from the lead investigator.
The Food Safety Technical Assessment Unit will carry out one of the following actions:
a) Provide estimate of the level of risk
The technical risk assessor evaluates the preliminary evidence available, taking into consideration the potential hazard, exposure and historical data, to provide an estimate of the level of risk. This estimation is provided for prioritization and to determine appropriate operational activities and response.
b) Provide a technical risk assessment
When a risk assessment is required for the purpose of the CFIA making a recall determination (3.1.1 b), or to determine whether a product action taken by a regulated party is sufficient to mitigate risk (3.1.1 c), the Food Safety Technical Assessment and Analysis Unit will triage the request according to established procedures to determine whether the risk can be assessed using a technical risk assessment or if it requires a health risk assessment from Health Canada. The decision to proceed with a technical risk assessment may be taken when the technical risk assessment is well supported by:
- a Health Canada guideline, policy or standard
- an existing incident-specific health risk assessment from Health Canada for a very similar situation (that addresses exact or similar situations) and where a similar set of assumptions were used
- a health risk assessment obtained for the same incident where it can be logically extended to other food identified in the food safety investigation (for example, when assessing the risk posed by a food made using a recalled ingredient or that has additional flavours, sizes or lots)
When it is determined that the risk will be assessed through the development of a technical risk assessment, the technical risk assessor will proceed with its development in consultation with Policy and Programs Branch, Science Branch or Health Canada as appropriate and according to established procedure (for example, to seek clarification on relevant guidelines, policies or standards, or for technical support when required
c) Prepare a request for a health risk assessment
When it is determined that a health risk assessment from Health Canada is required to assess the risk associated with an incident, the Food Safety Technical Assessment and Analysis Unit will prepare a formal request. The technical risk assessor will inform the food safety recall specialist that this approach was selected (refer to sections 3.2, 3.3 and 3.4).
3.2 Request a health risk assessment
When a decision to request a health risk assessment from Health Canada is made, the technical risk assessor will submit the request according to established procedures ensuring that Health Canada is provided with the information to support the risk assessment process. The Food Safety Technical Assessment and Analysis Unit will follow the protocol described in their operating procedures.
3.3 Conduct a health risk assessment
Health risk assessments are conducted for the CFIA by Health Canada. According to Codex Alimentarius, a risk assessment is a scientifically-based process consisting of the following steps: hazard identification, exposure assessment, hazard characterization and risk characterization. The health risk assessment normally concludes with a risk classification.
The health risk assessment process may trigger the need to seek additional information and/or clarifications from the OFSR that in turn may need to ask the Lead Investigator to gather further information. Please refer to Section 2.0 – Conduct a food safety investigation.
3.4 Receive the health risk assessment
Upon receiving the health risk assessment from Health Canada, the technical risk assessor will review it to ensure all information is correct and that it adequately addresses the request. The health risk assessment can be compared with existing documents pertaining to similar situations for consistency purposes. If clarifications are needed or discrepancies are identified, the technical risk assessor will bring them to the attention of the Health Canada assessor for clarification and resolution.
3.4.1 Risk classifications
Responses to risk assessment requests are conveyed by email from the Food Safety Technical Assessment and Analysis Unit to the Food Safety Investigation and Recall Unit. This response provides all relevant information, documents and/or references that support the identified risk classification.
The following are possible risk classifications:
- Health Risk 1 (HR1): The health risk identified represents a situation where there is a reasonable probability that the consumption/exposure to a food will lead to adverse health consequences which are serious or life- threatening, or that the probability of a foodborne outbreak situation is considered high.
- Health Risk 2 (HR2): The health risk identified represents a situation where there is a reasonable probability that the consumption/exposure to a food will lead to temporary or non-life threatening health consequences, or that the probability of serious adverse consequences is considered remote.
- Health Risk 3 (HR3): This represents a situation where there is a reasonable probability that the consumption/exposure to a food is not likely to result in any adverse health consequence. The situation identified may be an indication of a breakdown in Good Manufacturing Practices; in Good Agricultural Practices; in Good Practices in Veterinary Medicine or some other relevant factor.
There are other possible outcomes of a health risk assessment:
- The situation is not considered to represent a health risk; or
- Unable to assess: There is insufficient information for one or more steps of the risk assessment process to assign a health risk
4.0 Make a decision
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The fourth step, make a decision, is highlighted.
Overview
When a food is assessed as representing a health risk, the next step is to determine whether risk mitigation actions are necessary and which ones would be the most appropriate. This determination is based on the risk classification and other relevant factors. The risk mitigation response should be proportional to the risk and consistent across similar incidents.
The Office of Food Safety and Recall (OFSR) manages the food recall process and is responsible for identifying situations where requests for voluntary food recalls should be made to regulated parties on the basis that they represent situations where the Minister could issue a recall order as per section 19 of the Canadian Food Inspection Agency Act.
The following describes the 3 main activities that are part of the decision making process to mitigate risk associated with food in distribution and refers to other appropriate guidance for determining other types of risk mitigation actions such as licence suspension or cancellation, seizing or detaining food, etc., which may need to be considered to address the incident as a whole.

Description of Figure 5: Make a decision
Figure 5 is a flow chart that outlines the 3 sub-steps within the section "make a decision". The sub-steps are: review the risk assessment; identify risk mitigation options; and develop the risk mitigation plan.
4.1 Review the risk assessment
When the food safety recall specialist receives the risk classification from the Food Safety Technical Assessment and Analysis Unit, they will determine the appropriate next steps based on the outcome and other relevant factors. The possible outcomes are as follows:
- The risk classification indicates a risk that may need to be mitigated by a food recall
- The risk classification or health risk assessment may indicate that other risk mitigation actions may need to be considered, other than or in addition to a food recall
- The risk classification or elements of the health risk assessment may indicate that more risk evidence is needed and the food safety investigation should continue
- The risk classification indicates a level of risk that does not require further risk mitigation actions and the OFSR can close the file
4.2 Identify risk mitigation options
Based on the risk classification, the OFSR will determine if a food recall is a necessary risk mitigation action and whether a public warning needs to be issued. The lead investigator with the food safety investigation team, as appropriate, will identify other necessary measures to mitigate the risk posed by affected food remaining in inventory and to prevent reoccurrence or similar incidents from occurring in the future. These other measures, such as seizure, detention or product disposition, may be taken at any stage of the food safety investigation.
Refer to section 4.2.8 for a list of other risk mitigation actions.
4.2.1 Recall determination
A food recall is the removal of a food from further sale or use, or the correction of its label, at any point in the supply chain (for example, distributors, retailers, consumers, etc.) as a risk mitigation action.
After receiving the risk classification from the Food Safety Technical Assessment and Analysis Unit, the Food Safety Investigation and Recall Unit determines whether requesting a recall is the appropriate risk mitigation action for food that is in distribution. The determination is based on the level of risk posed by the food and other relevant factors, such as the shelf life of the food, the volume of food in distribution and the availability of the food in consumers' homes (for example, a refrigerated food that may have been frozen by a consumer).
In general, when a food is assessed as representing a Health Risk 1 or 2, the Canadian Food Inspection Agency (CFIA) will typically request a voluntary food recall.
When a food in distribution is assessed as representing a Health Risk 3, the CFIA will typically not request a recall. However, in the following Health Risk 3 situations a recall is requested:
- Priority allergens, where:
- there has been a reported reaction, or
- a Health Risk 3 was assigned and:
- the level of allergen can trigger a reaction, but was mitigated solely on the basis of low volume of distribution; or
- there is at least one compounding factor (for example, an allergen free claim)
- Non-priority allergens where there has been a reported reaction
- Where the area of concern is related to a microbial pathogen that is not likely to be mitigated by the consumer
- Where the area of concern is related to a parasite or virus and there is an outbreakFootnote 16
- Incident involving food targeted to infants and toddlers
- Where the food contains a veterinary drug that is prohibited under Section B.01.048 of the Food and Drugs Regulations
- The presence of spoilage, including rancidity, or contamination of the food with foreign material or insect/rodent infestation is evident and not considered to be isolated
- Foreign product action by legal agent and distribution to Canada is listed on their public communication
- Exceptional circumstances determined on a case-by-case basis by OFSR management (examples include, food targeted to vulnerable populations; company decides to do a public recall, seasonality)
Please refer to Appendix 4, Table 1 for more detailed information about risk mitigation options for Health Risk 3 situations.
Whether or not a food recall is requested, other risk mitigation actions may also be required to address food remaining in the regulated party's inventory and future production. The lead investigator should also follow any applicable guidance relevant to the incident.
For information on product disposition, refer to Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687).
4.2.2 Recall classification
The recall classification is the numerical designation (Class I, Class II or Class III) assigned by the OFSR to a particular food recall to indicate the relative degree of health risk represented by the food being recalled.
There are three recall classifications, which are typically based on the level of health risk posed by a food:
- Class I recall: the recalled food is assessed as representing a Health Risk 1.
- Class II recall: the recalled food is assessed as representing a Health Risk 2.
- Class III recall: the recalled food is assessed as representing a Health Risk 3.
4.2.3 Determination of the recall depth
After determining that a food recall is the appropriate risk mitigation action, the Food Safety Investigation and Recall Unit will determine the required depth of the recall. The depth of a recall describes the extent to which the recall is to be conducted into the food distribution system. The determination takes into consideration the type of clients who received the food (for example, consumers, retailers, institutions, etc.) and how far along the supply chain the food has been distributed.
The different depths of a recall are:
- Consumer level: Food is recalled from all levels of the supply chain up to the consumer level. This includes food sold only to consumers directly through internet sales.
- Hotel/Restaurant/Institution level: Food is recalled from all levels of the supply chain up to the hotel/restaurant/institution level.
- Retail level: Food is recalled from all levels of the supply chain up to the retail level (that is, food that is available on retail shelves).
- Warehouse/distribution level: Food is recalled from all levels of the supply chain up to the warehouse/distribution level.
The food safety recall specialist will use the following factors to determine if the recall should be conducted to the consumer level:
- The class of the recall
- Whether the food may have reached the consumer level
- Whether any illnesses, injuries or reactions associated with the food's consumption have been reported to the CFIA
- Whether the food is targeted at a vulnerable population, such as the elderly, infants, pregnant women and the immunocompromised
- The likely availability of the food in consumers' homes (for example, the food is sold refrigerated with a limited shelf life but it may have been frozen by the consumer)
- Whether the recalling firm or foreign food safety authority has issued or is planning to issue its own public warning
All Class I recalls are conducted to the greatest extent of distribution possible for the food in question (that is, either the consumer or hotel/restaurant/institution level, depending on the situation).
Class II recalls are conducted to the retail level or the hotel/restaurant/institution level as appropriate.
However, a Class II recall to the consumer level may be requested for the following situations:
- Ready-to-eat food contaminated with a bacterial pathogen (for example, Salmonella)
- An allergic reaction or illness was reported
- The food is targeted to infants/toddlers
- At the recalling firm's request
- The food is associated with a festive event or occasion
- Direct sale to the public (such as door to door, school fundraiser or internet sales)
- Foreign country initiated recalls where a warning is issued
- Based on action taken by, or at the request of, other government departments or
- Exceptional circumstances
Class III recalls are conducted to the retail or hotel/restaurant/institution level, as appropriate, with the exception of the following situations where a recall to the consumer level may be requested:
- the food is targeted to infants/toddlers
- foreign country initiated recalls
- foreign manufacturer's recall already in the public domain
- at the request of the recalling firm
- direct sale to the public (such as, door to door, school fundraiser or internet sales)
- any other exceptional circumstances
4.2.4 Recall communication
When a recall to the consumer level is conducted, a Food Recall Warning is often issued by the CFIA as part of the risk mitigation plan advising consumers not to consume a recalled food.
However, when a recall to the consumer level is conducted for food that was sold only to consumers directly through internet sales, a Food Recall Warning may not be issued if the recall does not meet the criteria for issuing one (for example, a Class III recall to consumer level).
A Food Recall Warning is a warning issued to consumers advising them not to consume a recalled food. When the hazard is an allergen, the Food Recall Warning is issued to advise allergic consumers.
A Food Recall Warning may be updated when additional information (for example, expanded food or distribution information, additional recalled foods, etc.) has been identified during a food safety investigation.
Food Recall Warnings are disseminated to the media by the CFIA.
For foodborne outbreak situations, the CFIA will reference public health notices issued by the Public Health Agency of Canada, as appropriate.
A recalling firm may decide to issue a public release regardless of the level of risk associated with the food being recalled. The CFIA will issue a warning as well when a food recall warning is necessary according to CFIA policy. Refer to Table 2: Summary of product action communication activities.
All recalls are posted on the Recalls and safety alerts website. Where a Food Recall Warning is issued or when the recall is due to an undeclared or incorrectly declared allergen an e-mail notification is also sent to subscribers and the recall is posted on the CFIA's social media channels.
There are some circumstances where the CFIA cannot issue a Food Recall Warning, but there is still a need to communicate the risk to the public. A Food Safety Warning may be issued by the CFIA to inform consumers of a food safety incident where a recall has not yet been conducted or cannot be conducted.
One of the following criteria has to be met before the recall specialist and/or national manager approaches the director of the OFSR with a recommendation to issue a Food Safety Warning. The decision to issue a Food Safety Warning is made by the director of the OFSR.
Food Safety Warnings can be issued for situations where the request would have been for a recall to the consumer level (any class) or a Class I or II recall to the HRI/retail levels.
The criteria are as follows:
- Company is not reachable within 2 hours of the lead investigator initiating contact, after all reasonable attempts by telephone and email have been made; or
- Company refuses to conduct the recall without delay and the level of risk is such that risk communication cannot wait; or
- The recalling firm has not been identified and it is not clear whether a recall has been initiated.
Please refer to Appendix 4, Table 2 for a summary of product action communication activities.
4.2.5 Product action initiated by regulated parties
When the regulated party has informed the CFIA that they have removed food from sale or corrected the label of a food along the supply chain based on their own risk assessment, the lead investigator will request a copy of the regulated party's assessment, if available and provide it to OFSR for consideration. The OFSR will determine whether the action taken by the regulated party was sufficient to mitigate the risk, which may involve requesting a risk classification where needed. In the event the regulated party's actions were not sufficient, the OFSR will inform the lead investigator of the appropriate risk mitigation actions required (refer to section 3.1.2. b).
Where the food represents a health risk, the recalling firm's action is classified according to the rationale outlined in section 4.2.1. The situation will be reviewed by the OFSR to determine if further risk mitigation actions are necessary, such as whether a public warning needs to be issued. If the recalling firm's action was appropriate for the level of risk, it is referred to as a company initiated recall.
In situations where the food does not represent a level of health risk and/or conditions that meet the CFIA's criteria for requesting a recall, but is in contravention of legislation administered or enforced by the CFIA, the action taken by the regulated party will be classified as a market withdrawal.
Situations where the action taken by the regulated party on a food that is in contravention of legislation administered or enforced by the CFIA, but has not left their direct control and, therefore, is not in the marketplace, will be classified as a stock recovery.
Situations where action has been taken by the regulated party on a food that is not in contravention of legislation administered or enforced by the CFIA will be classified as a product withdrawal. Most product withdrawals are for quality reasons.
The 3 classifications outlined above are not considered to be food recalls by the CFIA and are not posted on the Recalls and safety alerts website.
4.2.6 Recalls triggered by foreign parties
When the CFIA is informed of a food recall by a foreign company or food safety authority, the OFSR will confirm if the food has been distributed in Canada and classify the recall according to the information provided and the nature of the hazard, as well as Canadian standards and policies.
If the foreign recall is already in the public domain (in other words, a recalling firm has issued a public notice to the media or on its own website or the recall has been publicized by a foreign food safety authority) and it is confirmed that the food has been distributed in Canada and the importer(s) is known, the CFIA will issue a public warning. If the information is not available, the CFIA may issue a Food Safety Warning until such time that the Canadian importer(s) can be identified and the recall process can be initiated.
4.2.6.1 Non-resident importers
A non-resident importer (NRI) is a regulated party that holds a SFCR licence to import food into Canada whose fixed place of business is located in a country other than Canada.
When an incident involves food imported by a NRI, the CFIA will work with the NRI during the food safety investigation to verify whether a contravention or a hazard exists and to determine the nature and extent of the problem. The CFIA will ask the NRI to recall the food when a food imported by a NRI poses a health risk. However, as anyone involved in the distribution, marketing or sale of a food that poses a health risk can be asked to conduct a recall of that food and be named as the recalling firm, there may be situations where the Canadian distributor of the imported food is asked to conduct its recall. In determining the recalling firm, the CFIA will consider the facts and circumstances relating to the particular need for a recall.
4.2.7 Other risk communication actions
When a food recalled in Canada was imported or was exported outside of Canada, the OFSR will inform the implicated countries and, as appropriate, the secretariat of the International Food Safety Authorities Network.
There may be situations where a food recall is not conducted, but other risk communication action may be necessary.
A Consumer Advisory may be issued by the CFIA to inform consumers of an ongoing food safety investigation or to provide food safety recommendations in relation to an incident (for example, food tampering situations or to address concerns raised by the public).
Refer to Appendix 2, Procedure 8: CFIA guidelines for early public communication of food safety incidents for more information.
4.2.8 Other risk mitigation actions
When a food has been assessed as representing a risk or is in contravention of legislation, irrespective of whether a recall is requested, other risk mitigation actions will be used by the CFIA to address any food remaining in the regulated party's inventory and future production/imports. Risk mitigation actions can be taken at any time during the food safety investigation and recall process. More than one action can be implemented singularly or simultaneously, as needed.
Immediate and long-term options include, but are not limited to the following:
- Letter of non-compliance
- Seizing or detaining food or equipment
- Stopping production
- Increased inspection and sampling frequency
- Border lookouts
- Licence or registration suspension/cancellation
- Prosecution
- Develop fact sheets
- Review and update or develop guidelines/policies/regulations
- Industry sector audits
- Industry sector education
- Additional media relations and outreach activities to targeted groups
The lead investigator should also follow any applicable guidance relevant to the incident as cited in the Food Manual.
4.3 Develop a risk mitigation plan
A risk mitigation plan outlines the risk mitigation decision and activities that will be conducted by the CFIA and/or the regulated party to address a food safety incident. It can be written or verbal and scaled appropriately based on the size and/or complexity of the incident. It may be modified as necessary if new information becomes available.
Whether or not a recall is requested, other risk mitigation actions may be required. In such cases, a risk mitigation plan can be developed by the lead investigator and/or Food Safety Investigation Team, in collaboration with their supervisor and the area/regional recall coordinator, as appropriate, according to:
- regulatory requirements
- operational guidance
- advice from the OFSR and other advisory functions
The lead investigator can use control, enforcement or compliance actions to mitigate the risk. If the lead investigator determines that other actions are required in conjunction with control actions, they will refer to the appropriate operational guidance.
5.0 Mitigate the risk
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The fifth step, mitigate the risk, is highlighted.
Overview
When a decision is made with regard to risk mitigation actions, the lead investigator will communicate it to the regulated party for implementation. In the case of a recall determination, the Office of Food Safety and Recall (OFSR) will communicate the determination to the lead investigator and both the food safety recall specialist and the lead investigator will be involved in the implementation. The following figure outlines the steps involved in communicating the decision and implementing and verifying the effectiveness of the risk mitigation actions.

Description of Figure 6: Mitigate risk
Figure 6 is a flow chart that outlines the 4 sub-steps within the section "mitigate the risk". The sub-steps are: communicate the risk mitigation plan; implement the risk mitigation plan; verify completeness and effectiveness of actions; and conduct follow-up activities.
5.1 Communicate the risk mitigation plan
5.1.1 Notify the lead investigator
The food safety recall specialist will communicate to the lead investigator and/or the food safety investigation team the risk mitigation decisions related to food in distribution. Where it has been determined that a recall is a required risk mitigation action, the following is communicated to the lead investigator:
- The rationale for the recall determination
- The recall classification
- Whether a Food Recall Warning will be issued and the information in the Food Recall Warning to be shared with the regulated party
- The applicable legislation, where appropriateFootnote 9, that the food may be in contravention of with a recommendation that the inspectorate make the final determination
5.1.2 Communicate the risk mitigation decision to the regulated party
The lead investigator will communicate the recall determination to the regulated party along with other risk mitigation actions deemed necessary to address all aspects of the incident. They will ensure that the regulated party understands the basis for the recall request, where appropriate. As per area protocol, the lead investigator may consult with their supervisor and/or inspection manager before informing the regulated party.
Irrespective of the risk assigned to the food, a regulated party responsible for the food in question may wish to review details of the health risk assessment or the risk classification to assist them in reaching an agreement to conduct a voluntary recall. In this case, the lead investigator will follow Appendix 2, Procedure 7: Responding to a company's request for a copy of a risk assessment for food product.
Where the determination is to request a voluntary food recall, the lead investigator will communicate the request along with the following information to the regulated party:
- The rationale for the recall determination
- The recall classification
- Whether a Food Recall Warning will be issued and the information in the Food Recall Warning to be shared with the regulated party
- The section of the applicable legislation that the food contravenes, where appropriate
- Advise that the recall will be posted on the Recalls and safety alerts website
- The steps to be followed for an effective recall. Refer the regulated party to the Recall procedure: a guide for food businesses.
If the regulated party does not agree to conduct a voluntary recall or requires additional clarification regarding the request for a recall, the lead investigator will inform their supervisor, the area/regional recall coordinator and the OFSR.
If the regulated party agrees to conduct a voluntary recall, the lead investigator will send a confirmation letter to the recalling firm outlining their agreement to conduct the voluntary recall (refer to Appendix 3, Template 2: Standardized CFIA recall notification letter to recalling firm).
For recalls that are conducted to the consumer level and where a Food Recall Warning is required, the OFSR will draft the Food Recall Warning in the Recalls and Safety Alerts System (RSAMS). The OFSR will share a product table listing the products that are subject to recall, distribution information, and the company's name with the lead investigator via email, who will consult with the implicated recalling firm to ensure the accuracy of the food's description, the distribution information as well as the recalling firm's name. Once finalized, the OFSR will post the Food Recall Warning on the Recalls and safety alerts website, and Communications and Public Affairs Branch will post it on the CFIA's social media channels. The Agency standard is to issue a Food Recall Warning within 24 hours of the recall determination.
The issuance of a Food Recall Warning may be expedited under specific circumstances outlined in Procedure 6: Expediting the issuance of a Food Recall Warning when, based on preliminary risk analysis, the CFIA has sufficient confidence that a warning will be necessary and that it would need to be issued the same day. In these situations, the CFIA may share pertinent information from the draft Food Recall Warning with the implicated regulated party either before a health risk assessment is obtained and/or before the final risk mitigation decision is made.
In situations where a recall is not considered as a necessary risk mitigation action, the lead investigator will communicate the information to the regulated party. Irrespective of whether or not a food recall is required, the lead investigator will also communicate if any other relevant risk mitigation actions need to be taken.
5.1.3 Communicate the risk mitigation decision to the inspector responsible for overseeing the regulated party
The lead investigator will communicate the recall to the inspector responsible for overseeing the regulated party so that they can initiate a preventive control inspection, as appropriate. If the lead investigator is also the inspector responsible for the regulated party, they will initiate the preventive control inspection in response to the recall.
For more information, please refer to the Food preventive control and traceability inspection – Compliance verification system guidance.
5.1.4 Mandatory recall order
If the regulated party is not willing to conduct a voluntary recall of the affected food, the CFIA may consider seeking a recall order from the Minister of Health. Recall authorities are provided under section 19 of the Canadian Food Inspection Agency Act.
Please follow Appendix 2, Procedure 9: Mandatory recalls for the procedure.
Other situations where seeking a mandatory recall order may need to be considered include when the responsible regulated party is not known or is unable to conduct a recall (for example, the regulated party is no longer in business).
Alternatively, the CFIA may decide to approach clients who received the affected food, such as distributors, retailers, etc., to request a voluntary recall.
5.1.5 Recall of food representing various levels of health risk
In some situations, a recall may involve multiple foods assessed as representing different levels of risk. In such a case, the CFIA will document the highest level of risk and depth of the recall in the Issues Management System (IMS). It is the CFIA's policy to include all of these foods on one public warning or recall posting (notification) to avoid confusion for the public and industry.
5.2 Implement the risk mitigation plan
5.2.1 Assisting the recalling firm's recall implementation
The recalling firm is responsible for the implementation of the food recall. The lead investigator will assist the recalling firm by obtaining and reviewing the following information:
- A copy of their notice of recall, which includes the recalling firm's product disposition instructions
- The recalling firm's own public release, if applicable
- The distribution list specific to the recalled food
5.2.2 Internal and external stakeholders
When there is a recall that requires a public warning, the OFSR will share the final public warning with the following internal stakeholders:
- Office of the President
- Web Publishing Unit
- Area/regional recall coordinators
- Operations Policy and Systems Directorate
- National Issues Management Office
- International Affairs and Market Access
- Food Import and Export Division
The OFSR also shares the final public warning with the following external stakeholders and other government departments:
- Canadian Federation of Independent Grocers
- Retail Council of Canada
- National Foodservice Distributors Association
- Canadian Produce Marketing Association
- The Public Health Agency of Canada
- Health Canada
- The Privy Council Office
5.2.3 International notification of food recalls and other safety concerns
Canadian food recalls may involve imported food or food that has been exported to other countries. The OFSR will share information on recalls with foreign food safety authorities when food recalled in Canada was either exported to them or imported from them via:
- member countries of the International Food Safety Authorities Network through their National Emergency Contact Point
- non-members of the International Food Safety Authorities Network through the International Affairs and Market Access Office via the appropriate embassy contact points
- food recalls that only involve the USA (no other trading countries), OFSR informs the appropriate US food safety authority of all recalls where the recalled food either originated in the USA or was exported to the USA
The OFSR will also notify the International Food Safety Authorities Network Secretariat of all Class I and II recalls of food that entered international trade with the exception of allergen-related recalls and recalls involving food imported from or exported to the United States of America only.
5.2.4 Notice of recall
For all classes of recalls, the recalling firm will notify each of its affected receiving clients without delay. The purpose of a recall notice is to:
- notify the clients who may have received the recalled food of the recall
- prevent the further sale, distribution or use of the recalled food
The lead investigator will review the notice of recall to ensure that it:
- clearly identifies the food, package size, lot number(s), package and case code(s), and/or UPC codes, etc.
- explains the reason for the recall and the hazard involved
- provides specific instructions on product disposition
- provides specific instructions on acknowledging the receipt of the Notice of Recall
- clearly states that the receiving clients should, in turn, notify any of their clients to whom they further distributed the food that a recall is being conducted, where appropriate
- does not contain irrelevant qualifications, promotional materials, or any other statement that may diminish the recall message
Receiving clients will conduct the recall and notify their receiving clients if they have further distributed the recalled food. Industry, at every level of distribution, will ensure the recalled food is removed from sale or further use according to the depth of the recall.
Industry can refer to the Appendix 3, Template 4 for an example of a recall notice template.
5.2.5 Product disposition
The lead investigator will:
- request and review the recalling firm's method of product disposition that will be used by receiving clients
- ensure the product disposition method is appropriate and validated
For additional guidance regarding disposition procedures, please follow Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687).
5.2.6 Other risk mitigation actions
Where other risk mitigation actions are required, the lead investigator will:
- ensure that the recalling firm understands the request
- ensure the recalling firm corrects the incident that led to the unsafe food
- ensure appropriate corrective actions are in place and implemented to prevent similar incidents in the future
A list of other risk mitigation actions can be found in section 4.2.8.
For information on product disposition, please refer to Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687).
Please refer to the Standard regulatory response process to determine appropriate compliance actions, including enforcement actions.
The lead investigator can consult with their supervisor and any other inspectors or specialists, who have appropriate experience and knowledge, as needed.
5.3 Verify completeness and effectiveness of recall actions
Recall verification is the process by which the CFIA determines whether the recalling firm has been diligent and successful in identifying the affected food; notifying all affected receiving clients of the recall; and that the receiving clients have responded accordingly to the notification by taking the appropriate product action.
Refer to Appendix 2, Procedure 1, Table 3 for a summary of the main steps and activities to be conducted.
5.3.1 Recall verification
The objectives of the recall verification activities are to evaluate the:
- overall completeness and effectiveness of the recall
- recalling firm's recall process
- actions taken by the receiving clients when advised of the recall
- actions taken when deviations are observed
Verification of the completeness and effectiveness of Class I and II recalls are conducted by following these two approaches:
- A review of the results of the recalling firm's recall activities (refer to section 5.3.1.1) and
- Verification of the receiving clients' actions through the use of a statistical approach (refer to section 5.3.1.2)
Generally, for Class III recalls, verification will be limited to the review of the results of the recalling firm's activities; however, there may be exceptional circumstances where the second approach may be required (refer to section 5.3.1.2).
For complete details on the recall verification process, please refer to Appendix 2, Procedure 1.
The CFIA will take appropriate actions (for example, request that the recall be re-done) should it be determined that the recalling firm has not been successful in conducting an effective recall.
When verifying client accounts during recall effectiveness checks, inspectors may become aware that a recalled food and/or ingredient was sold to a cannabis licence holder. The inspector will notify the OFSR and, in turn, the OFSR will notify Health Canada so they can take appropriate action in respect of the finished cannabis product.
5.3.1.1 Review of recalling firm's activities
For all recalls, the lead investigator will verify the effectiveness of the recall by conducting a review of the results of the recalling firm's recall activities. The document review will be conducted with the recalling firm in accordance with the established time frames for verification activities (refer to Appendix 2, Procedure 1, Table 1).
5.3.1.2 Verification of receiving clients' actions
For all Class I and II recalls, the lead investigator and the assigned inspectors will verify the actions taken by the recalling firm's clients. This will be done by using a statistical approach where a certain number of receiving clients will be selected for verification. For the verification procedure, please refer to Appendix 2, Procedure 1.
Verification of the actions taken by the clients is typically not necessary for Class III recalls; however they may need to be conducted for the following Class III situations:
- a recall of a food intended for a vulnerable population
- when a public warning has been issued by the CFIA
- when a regulated party (recalling firm or receiving client) has a history of non-compliance
- exceptional circumstances
For any class of recall, verification of the clients' actions may need to be modified depending on the situation. For example, it may be necessary to perform verifications with 100% of the receiving clients when the recalled food is distributed to vulnerable populations. Likewise, the percentage may be reduced when CFIA has sufficient confidence and evidence that the recalled food has been fully removed from the market. Before modifying the verification approach, the lead investigator will consult with their supervisor and/or inspection manager, the area/regional recall coordinator and the OFSR. Refer to Appendix 2, Procedure 1.
When sending a request for effectiveness checks, the lead investigator will include a brief rationale and, when relevant, an explanation as to why the approach is modified. Assigned areas/regions will conduct the verification activities as requested.
The lead investigator will document the decision, and when applicable the modified method used, in the IMS. Refer to Appendix 2, Procedure 5: Documenting key decisions during food safety investigations and recalls for additional information.
5.3.1.3 Time frames for initiating and reporting recall verification activities
Recall verification activities should start and be completed within a specified number of business days after the initiation of a recall as shown in Appendix 2, Procedure 1, Table 1.
When recall effectiveness checks are conducted by another government authority on the CFIA's behalf (for example, in Quebec, the ministère de l'Agriculture, des Pêcheries et de l'Alimentation du Québec conducts recall effectiveness checks on the CFIA's behalf), the time frames for completing the verification activities may need to be adjusted depending on the applicable Memorandum of Understanding and/or the operational needs of the other government authority.
5.3.1.4 Verification of recalls triggered by a foodborne illness outbreak
Verification activities for recalls triggered by a foodborne illness outbreak will be given priority over verification activities for all other recalls. The activities will begin immediately after the recall and the lead investigator will consult with the area/regional recall coordinator and the OFSR on the appropriate level of verification (for example, 100% verification checks may need be required for specific situations).
In the effectiveness check request, the lead investigator will emphasize the priority to be given. Assigned areas/regions will conduct the verification activities as requested.
5.3.1.5 Evaluation of the overall effectiveness of the recall
The lead investigator will evaluate the number and nature of the deviations encountered during the recall verification process and determine if the recall was effective or ineffective (refer to Appendix 2, Procedure 1). This evaluation will include the findings of the document review at the recalling firm together with the results of the verification activities conducted at their clients. If the lead inspector determines that the recalling firm has not been successful in conducting an effective recall, the recalling firm will be requested to take appropriate actions to ensure the safety and well-being of consumers.
5.4 Conduct activities following incident response actions
When a food has been assessed as representing a risk or is in contravention of legislation, irrespective of whether a recall was requested, actions should be taken by the responsible regulated party to prevent reoccurrence of the incident by implementing appropriate corrective actions and ensuring the appropriate disposition of the affected food.
These activities will target food that is not on the market including:
- recalled food returned to the recalling firm
- affected food that had not left the responsible regulated party's control
- new production or imports
The lead investigator will verify if the following corrective measures were implemented and effective:
- recovery, reconciliation, control and disposition of the recalled food
- control and disposition of affected food that has not left the recalling firm's control
The inspector responsible for conducting activities following an incident response will verify that the following corrective measures were implemented and effective:
- appropriate action to correct the root cause of the incident
- the recalling firm's Preventive Control Plan is revised, as required
and will communicate their findings to the lead investigator.
If a new hazard is identified during these activities, the Food incident response process will be re-initiated.
5.4.1 Product disposition
The lead investigator will assess, monitor and document disposition activities undertaken by the responsible regulated party as well as at locations where the affected food is not under the control of the responsible regulated party. There are various methods of disposition that can be used by the responsible regulated party:
- Relabeling, reworking, inspecting/culling or any other action to bring affected food back into compliance
- Exporting the affected food to a country willing to accept it, including food returned to the country of origin or
- Destruction/disposal of the affected food
The lead investigator will follow Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687).
5.4.2 Verification that the regulated party has addressed the root cause of the recall
The lead investigator will follow section 6.1.1 of the Food preventive control and traceability inspection – Compliance verification system.
5.4.3 Other activities
Other follow-up activities can include:
- providing advice and recommendations to Policy and Programs Branch that can help in the review of standards/policies
- collaborating with industry sectors or other countries to address trends affecting more than one firm or sector
For additional information and procedures on activities following incident response actions, please follow the Food preventive control and traceability inspection – Compliance verification system.
Enforcement or compliance actions can continue after the risk is controlled. If enforcement actions are required, please refer to the Standard regulatory response procedure: control and enforcement (accessible only on the Government of Canada network – RDIMS 9146498) and/or to the Food regulatory response guidelines.
When a food does not represent a health risk, but there is a non-compliance, the lead investigator will take appropriate compliance actions according to the Standard inspection process and/or other appropriate guidance.
6.0 Close the incident file
Description of Figure 1: Food incident response overview
Figure 1 is a flow chart that outlines the 6 steps of the food incident response process: conduct a preliminary assessment; conduct a food safety investigation, assess the risk, make a decision, mitigate the risk and close the incident file. The sixth step, close the incident file, is highlighted.
Overview
In order to close the incident file, the lead investigator will verify that all relevant information, including the rationale for closing the incident file, has been appropriately documented in the Issues Management System (IMS) and that all of the steps required for closing the file have been followed.

Description of Figure 7: Closing the incident file in the IMS
Figure 7 is a flow chart that outlines the 5 sub-steps within the section "close the incident file". The sub-steps are: develop a rationale; seek approval; demobilize resources; conduct lessons learned; and communicate the incident file details.
6.1 Develop a rationale
The lead investigator will decide when the incident file should be closed in the IMS when the following applicable conditions have been met:
- No potential health risk or contravention has been identified
- The health risk has been mitigated
- The food was recalled and/or other risk mitigation actions have been taken
- Activities following incident response actions are opened/scheduled in the Digital Service Delivery Platform (DSDP)
- All reasonable lines of enquiry have been investigated
- Corrective actions have been taken to prevent reoccurrence
- The incident falls outside of the Control Function or the Canadian Food Inspection Agency (CFIA) mandate and needs to be referred
- All portions of the food safety investigation are completed and closed in the IMS by the Office of Food Safety and Recall (OFSR) and all assigned inspectors, as appropriate
- When the trigger was a complaint, the complainant has been contacted as per the Operational procedure: Responding to food complaints (accessible only on the Government of Canada network – RDIMS 13899359)
In some situations, the lead investigator may close the incident file before all the conditions are met and will include a rationale (for example, guidance is to be obtained from the Operational Guidance and Expertise Unit).
When closing the incident file, the lead investigator will ensure that all inspection documents related to the incident are recorded as per the Business rules of use for the IMS and various applicable procedures (accessible only on the Government of Canada network). The lead investigator will follow-up with the appropriate office to ensure information is entered accordingly, as necessary.
6.2 Seek approval
The lead investigator may consult with the area/regional recall coordinator and/or the OFSR before closing an incident file and may also seek approval from their supervisor, as per the Area protocol.
For guidance on how to close specific portions of the food safety investigation, please refer to Appendix 2, Procedure 5: documenting key decisions during food safety investigations and recalls.
When closing the incident file, the lead investigator will enter the deviation and the rationale for closing in the IMS.
For procedures on how to close an incident file in the IMS, please refer to the business rules of use for the IMS and the IMS user manual.
6.3 Demobilize resources
When an Incident Command System has been invoked, the emergency response teams will follow the food safety emergency response functional plan to demobilize the resources.
6.4 Conduct lessons learned
Depending on the nature and complexity of the food safety investigation or at the request of CFIA management, a debrief (also known as a "hot wash") can be conducted to identify which processes were effective and where improvements can be made. It is a critical step to ensuring continual improvement in the Agency's response to food safety incidents.
When an Incident Command System has been invoked, the emergency response teams will follow the food safety emergency response functional plan.
6.5 Communicate the incident file details
6.5.1 Internal stakeholders
Where control activities lead to the identification of a need for a policy or standard revision or with re-occurring food safety concerns or recalls, the incident is referred to the appropriate Centre of Operational Guidance and Expertise within the CFIA for action (for example, a letter to industry on appropriate controls, request to Health Canada to revise a policy or standard, etc.).
6.5.2 External stakeholders
Whenever there is communication with external stakeholders, the Protocol for CFIA's Sharing of Information during Food Safety Investigations and Recalls should be followed. Please also refer to the protocol for guidance on what type of information can be shared during food safety investigations and recalls, as well as the type of information that the CFIA is obligated to protect.
For certain situations described below, the CFIA will post a food safety investigation summary:
- The CFIA has issued a Class I recall with a public warning as a result of a foodborne illness outbreak in Canada or
- The Food Safety Emergency Response Functional Plan was activated
Appendix 1: Definitions
- Affected food
- Food identified through a food safety investigation as either posing a health risk or contravening legislation enforced by the Canadian Food Inspection Agency.
- Area of concern
- The specific nature of an incident (for example, undeclared allergens, microbiological contamination, chemical contamination, tampering, nutritional concerns, inaccurate labelling or net weight declarations, etc.).
- Control activities
- Activities undertaken by the CFIA to address a food incident.
- Consumer Advisory
- A general communication document to inform the public that the CFIA is aware of a particular situation and is taking appropriate actions. It may or may not provide food safety advice relating to a food and is typically used for situations where a health risk has not been established.
- Core business hours
- The fixed block of time during which CFIA employees are expected to conduct work in their respective time zones.
- Disposition
- Any action taken on an affected food to bring it into compliance or to remove it from the Canadian marketplace, such as relabelling, reworking, culling, exporting, destruction, etc.
- Early referral
- An immediate referral to the Office of Food Safety and Recall that is done during the preliminary assessment of a food safety incident or in the early stages of a food safety investigation, even though available information may still be limited. Please refer to Procedure 11, Annex A: High priority food safety incidents with requirements for early referral for the types of food safety incidents that require an early referral.
- Food
- Any article manufactured, sold or represented for use as food or drink for human beings, chewing gum, and any ingredient that may be mixed with food for any purpose whatever (see the Food and Drugs Act).
- Food emergency
- Any situation involving or potentially involving food which may pose a high health and safety concern to humans. Emergencies usually involve significant resources and require the coordination of a timely and/or extraordinary operational response.
- Food Recall Warning
- A warning issued to consumers advising them not to consume a recalled food as it represents a health risk. When the hazard is an allergen, the Food Recall Warning is issued to advise allergic consumers.
- Food safety incident
- A situation that has the potential to represent a health risk and that requires investigation under the CFIA's mandate.
- Food safety investigation
- An investigation into a food safety incident that includes inspection and related activities undertaken by regulatory officials to verify whether or not a food hazard, which could represent a health risk, exists and to determine the nature and extent of the problem, It is not a regulatory investigation which is conducted in response to non-compliance in order to recommend further compliance measures or enforcement action.
- Food Safety Warning
- A warning to consumers related to a food safety incident in which a recall has not yet been conducted or cannot be conducted (for example, when a recalling firm has not yet been identified).
- Health risk assessment
- A specific type of risk assessment conducted by Health Canada at the request of the Canadian Food Inspection Agency.
- High priority incident
- A food safety incident that takes precedence over all other work by Operations Branch. Please refer to Procedure 10, Annex A: High priority food safety incidents for a list of high priority food safety incidents.
- In distribution
- A food is considered to be in distribution when it has left the ownership of the regulated party responsible for its importation or manufacture.
- Mandatory recall
- Occurs when the Minister of Health orders a regulated party to recall a food that poses a risk to public health. A recall order may be issued in circumstances where a regulated party is unknown, unavailable or unwilling to conduct a voluntary recall, as per Section 19 of the Canadian Food Inspection Agency Act.
- Market withdrawal
- A regulated party's removal or correction of a marketed product that is in contravention of legislation administered or enforced by the CFIA, but does not represent a level of health risk and/or conditions that meet the CFIA's criteria for requesting a recall. It is not considered to be a recall.
- Normal priority incident
- A food safety incident that is not considered a high priority. Normal priority incidents do not appear in Procedure 10, Annex A: High priority food safety incidents.
- Notice of recall
- A notice that is issued by the regulated party to its receiving clients to inform them of a food recall, identifying the food to be recalled, the reason for the recall and providing instructions on its disposition.
- Outbreak
- An incident in which two or more people experience similar illness after a common source exposure. An outbreak is identified through laboratory surveillance or an increase in illness that is unusual in terms of time or geography. An outbreak is confirmed through laboratory and/or epidemiological evidence (Source: Canada's Food-borne Illness Outbreak Response Protocol).
- Overtime
- Authorized time worked by a Canadian Food Inspection Agency employee in excess of the standard daily or weekly hours of work and for which the employee may be entitled to compensation pursuant to the provisions of a collective agreement or CFIA policy. This may also be referred to as "after-hours work".
- Primary recall
- The first recall conducted during a food safety investigation. Also known as a recall incident.
- Product withdrawal
- A regulated party's removal from further sale or use of a marketed product that does not contravene legislation administered or enforced by the CFIA. It is not considered to be a recall.
- Public warning
- The communication document issued by the CFIA to inform the public of various health and safety concerns. Public warning documents include Food Recall Warnings, Food Recall Warnings (Allergen), Food Safety Warnings and General and Consumer Advisories.
- Recall
- The removal of a food from further sale or use, or the correction of its label, at any point in the supply chain as a risk mitigation action.
- Recall classification
-
The numerical designation (Class I, Class II or Class III) assigned by the Office of Food Safety and Recall to a particular food recall to indicate the relative degree of health risk represented by the food being recalled.
Class I represents a situation where there is a reasonable probability that the consumption/exposure to a food will lead to adverse health consequences which are serious or life- threatening, or that the probability of a foodborne outbreak situation is considered high.
Class II represents a situation where there is a reasonable probability that the consumption/exposure to a food will lead to temporary or non-life threatening health consequences, or that the probability of serious adverse consequences is considered remote.
Class III represents a situation where there is a reasonable probability that the consumption/exposure to a food is not likely to result in any adverse health consequence. The situation identified may be an indication of a breakdown in Good Manufacturing Practices; in Good Agricultural Practices; in Good Practices in Veterinary Medicine or some other relevant factor.
- Recall depth
- The level of distribution to which a recall is conducted (consumer, retail, hotel/restaurant/institution or wholesale).
- Recall verification
- The process by which the Canadian Food Inspection Agency verifies that the recalling firm has been diligent and effective in notifying their receiving clients of the need to recall the affected food and that the receiving clients have responded according to the notification.
- Recall incident
- A primary recall represents a breakdown in the food safety system leading to the request for a recall. A recall incident may lead to additional recalls (secondary) and have a common factor(s) such as product, cause, process deviation, etc. (for example, an out of country manufacturer recalls food that was shipped to five importers. This is captured as one recall incident (primary recall) and five recalls in total). The number of total recalls is the measure of all recalls conducted and includes recall incidents and secondary recalls. The number of secondary recalls varies by recall incident.
- Recall procedure
- A regulated party's procedure for effectively implementing a recall.
- Recalling firm
- A regulated party that is accountable for the implementation of a recall.
- Referral
- The step to formally engage the Office of Food Safety and Recall for coordination and guidance with respect to a food safety investigation or a recall.
- Risk classification
- The potential level of risk to Canadian consumers posed by a specific incident.
- Risk estimation
- An evaluation of the available information of a specific incident to determine the potential risk level for operational requirements.
- Root cause
- The original source of, or reason for, the food contamination or hazard (for example, inadequate sanitation, failure of process).
- Secondary recall(s)
- An additional recall(s) conducted during the same food safety investigation as the primary recall. The primary recall and secondary recalls have common factors, such as product, root cause, process deviation, etc. The trigger of secondary recalls is the primary recall incident.
- Stock recovery
- A firm's removal or correction of a product in contravention that has not been marketed and/or that has not left the direct control of the recalling firm. It is not considered to be a recall.
- Suspect food
- A food which triggers the food safety investigation or the food which through further food safety investigation has been considered to represent a risk.
- Technical risk assessment
- A science based process undertaken by the Canadian Food Inspection Agency (Office of Food Safety and Recall) which determines the risk associated with a food based on existing Health Canada guidelines, policies, standards or previous health risk assessments.
- Trigger
- Any specific source of information that prompts Canadian Food Inspection Agency staff to initiate a food investigation (for example, laboratory analysis, a consumer or trade complaint, a referral from another government department, an inspection finding, a report from a foreign government, a manufacturer-initiated product action or withdrawal, epidemiological evidence, etc.).
- Updated Food Recall Warning
- An updated warning that is issued when additional information concerning a food recall has been identified during a food safety investigation (for example, updated food or distribution information, additional food being recalled, etc.).
- Voluntary recall
- A recall that is initiated and carried out by the recalling firm without ministerial order.
- Vulnerable population
- A population that is at an increased risk for food-borne illness, such as the elderly, infants, pregnant women and the immunocompromised.
Appendix 2: Procedures
Procedure 1: Recall verification
When a food is subject to recall, the CFIA will conduct the following verifications to determine whether or not the recall was effective:
- A review of the results of the recalling firm's recall activities (refer to section 5.3.1.1) and
- Verification of the receiving clients' actions through the use of a statistical approach (refer to section 5.3.1.2)
These are the steps to be followed to determine the effectiveness of the recall.
Step 1: Request a detailed distribution list of clients from the recalling firm
The lead investigator will immediately ask the recalling firm to provide a detailed distribution list identifying the clients who received the recalled food.
Refer to Addendum 1: Recommended procedure for obtaining distribution list(s) of clients.
1.1 Review and verify the distribution list
The distribution list should include:
- the name and address (street address, unit number, city/town and province) for each receiving client
- the name, telephone number(s) and, if applicable, email address of the contact at each receiving client
- the type of client (for example, distributor, retailer, clients who serve vulnerable populations, such as a hospitals, care homes or daycares), including if the client is a licenced cannabis holder
- the amount of recalled food per lot code, when applicable, shipped to each client
If key information is missing from the distribution list, the lead investigator will follow-up with the recalling firm.
If a recalling firm is unable to provide the distribution list in a timely manner, there are a few options to keep the process moving:
- if available, the lead investigator may use a list from a previous recall conducted by the firm; although the distribution may have changed, this action expedites the process when current data is unavailable.
- the recalling firm may be able to provide a partial list from which the process may be started
1.2 Verify the type of clients
The lead investigator will review the distribution list.
- If the list contains distributors, the lead investigator will immediately send a request to the areas where the distributors are located so assigned inspectors can verify that all distributors were notified of the recall and obtain their list of clients. Refer to Step 4 for the details to be included in the request.
- If the list contains manufacturers, or retailers that have further processed/repackaged the recalled product, the lead investigator will initiate or notify the appropriate Canadian Food Inspection Agency (CFIA) offices to initiate additional food safety investigations, and will update their supervisor, area/regional recall coordinator and the Office of Food Safety and Recall (OFSR) as secondary recalls may need to be conducted.
- If the list contains foreign clients, the lead investigator will provide the contact information to the OFSR for notification of the respective foreign food authority. No further action will be taken on foreign client accounts.
- If the list contains clients who serve a vulnerable population, recall effectiveness checks (refer to Step 5) may need to be conducted for all of these clients. The lead investigator should consult with the area/regional recall coordinator and the OFSR for guidance. Refer to section 3.1.5.
- If the recalling firm distributed the food to fundraisers, recall verification should be done with these clients as fundraising organizations function in a similar manner to distributors. See Note.
- If the recalling firm sells food directly to individuals through the internet or has a loyalty or membership program, recall verifications should not be done with individuals even though the recalling firm is likely to have detailed contact information for each of their clients. See Note below.
- If the list contains clients who are cannabis licence holders, the lead investigator will send the client information and food information to the area/regional recall coordinator and the OFSR.
Note: As effectiveness checks are not required at the consumer level, the lead investigator and/or assigned inspector will not ask clients for the list of individual consumers who purchased the recalled food. However, these firms should be encouraged to notify their individual consumers of the recall, especially for recalls where a public warning is issued. Under certain situations, conducting effectiveness checks with individual consumers may need to be considered (for example, a recall involving food sold to a vulnerable population). The lead investigator will evaluate these situations on a case-by-case basis in consultation with their supervisor, area/regional recall coordinator and the OFSR, as appropriate.
1.3 Request a distribution list from the distributors
Upon receiving the request to contact distributors (refer to Addendum 3: Recall effectiveness check request template), the assigned inspectors will:
- review the recall notice
- determine if each distributor was notified of the recall, took appropriate product action, and notified their clients of the recall
- ask each distributor to provide a detailed distribution list of clients who received the recalled food or who were notified of the recall
- if there is no or very limited collaboration, the lead investigator or assigned inspector will conduct an onsite visit to request the distribution list
- follow steps 1.1 to 1.3 above with respect to each distribution list obtained
- keep the distribution lists until the recall verification plan is provided
Please refer to Addendum 1: Recommended procedure for obtaining distribution list(s) of clients.
Appendix 3, Template 5: Letter template to distributors requesting recall action is a tool which may be used as appropriate and in consultation with the supervisor and/or the lead investigator.
Data Entry:
- Assigned inspectors should record their activities in the Issues Management System (IMS) as a "Task" entry.
Step 2: Identify the appropriate recall verification plan to use
The lead investigator will identify the appropriate recall verification plan to be followed and will determine:
- the number of clients to select
- the sampling interval
- the random starting point
2.1 Determine the actual number or best estimate of clients
The lead investigator will remove any duplicate clients and foreign clients from the recalling firm's distribution list and will determine the actual number of receiving clients that are retailers, restaurants or institutions. The lead investigator should ensure that the OFSR has been informed about foreign clients so the respective foreign food authority may be notified. In addition, the lead investigator will exclude any distributors and fundraisers from the list of clients if they have already been verified. Refer to 1.2.e and 1.3.
Likewise, assigned inspectors liaising with the distributors will remove duplicate clients, foreign clients and clients that have already been verified from each client list. The inspector should ensure that the OFSR has been informed about foreign clients so the respective foreign food authority may be notified. The remaining number of clients on each list will then be provided to the lead investigator.
If complete client lists are not available in a timely manner, the lead investigator will collaborate with the recalling firm, and the assigned inspectors liaising with the distributors, to determine the best estimate of the number of clients who have or may have received the recalled food.
The best estimate can be determined via telephone or email without obtaining actual copies of the lists. However, care must be taken to ensure that the estimate does not differ significantly from the actual number of all clients excluding distributors.
Example
If there are multiple distribution lists involved, but not all have been received and the lead investigator needs to proceed with selecting the sampling interval and random starting point so that assigned inspectors that have received their entire distribution lists can proceed with the effectiveness checks, a best estimate can be used.
If the recalling firm has 50 retailers and 5 distributors and the 5 distributors in turn have approximately 400, 200, 300, 100 and 150 retailers, respectively, the best estimate of the number of clients is (50+400+200+300+100+150) 1200 retailers.
Data Entry:
- The lead investigator will record the actual/estimated number of clients in the IMS: "Recall tab, Eff. Check Summary tab, Number of Consignees tab, Total Number of Consignees".
2.2 Determine if the distribution is single or multi-channel
The lead investigator will determine if the distribution is single channel or multi-channel distribution.
| Single channel distribution | Multi-channel distribution |
|---|---|
| All clients are supplied by a single immediate supplier. | Not all clients have the same immediate supplier. Clients will be in two or more groups which are supplied by two or more immediate suppliers |
For a visual representation of these definitions refer to Addendum 2: Determining whether the distribution is single channel or multi-channel.
2.3 Determine the sample size and critical number
Knowing whether the distribution is single or multi-channel, the lead investigator will then determine the appropriate sample size and the corresponding critical number by:
- applying the recall classification and the type of distribution channel to Table 4: Selection of appropriate table for recall effectiveness checks to determine which recall effectiveness verification table (Tables 5-8) is appropriate for the recall effectiveness activities
- Using the identified sampling table, applying the actual or the best estimate number of clients to the first column and obtaining the corresponding sample size and critical number from the second and third columns, respectively.
Sample size is defined as the minimum number of clients where a recall effectiveness check will be conducted.
The critical number is defined as the maximum number of times the recalled product may be found being offered for sale, during the recall effectiveness check process, that does not render the recall as being ineffective. When the critical number is exceeded the lead investigator will typically determine the recall to be ineffective.
Example
For a class I recall with multi-channel distribution and a list of 1200 clients, Table 4 indicates that the appropriate recall effectiveness verification table to use is Table 5: Selection of clients for Class I multi-channel. The table indicates the sample size is 160 and the critical number is 7.
2.4 Determine the sampling interval
The sampling interval is used to select specific clients (sampling units) following a predetermined frequency (every Kth number) producing a random selection from the total list of clients (population).
The lead investigator will determine the sampling interval by dividing the actual number or the best estimate of clients by the sample size. If the calculated value is not a whole number, it will be rounded to the nearest whole number using mathematical rounding rules.
Example
For a class I recall with multi-channel distribution and a list of 1200 clients the sampling size is 160. From this the sampling interval is calculated to be 8 (1200/160 = 7.50, which is rounded to 8 to identify the clients in the list where effectiveness checks will be carried out).
2.5 Determine the starting point
The lead investigator will determine the starting point by randomly selecting a number between 1 and the sampling interval.
Example
For a class I recall with multi-channel distribution and a list of 1200 clients and a calculated sampling interval of 8, the random starting point is a number between 1 and 8 (refer to section 2.4). For example, with a randomly selected starting point of 3, the first client selected from each distribution list will be the third client listed.
Data Entry:
- The lead investigator will record the sample size and critical number in the IMS: "Recall tab, Eff. Check Summary tab, Number of Consignees tab", select appropriate recall effectiveness verification table from the drop down menu in the "Table Used" field. Enter the estimated number of checks to be carried out, critical number, starting number and interval in the appropriate fields.
2.6 Determine the number of clients to be selected in each distribution list
The lead investigator and/or assigned inspector will determine the number of clients that will be selected from each distribution list. To do this, divide the number of clients on each distribution list by the sampling interval which should contain two decimal places (to increase accuracy of the method) and round the result to the nearest whole number using mathematical rounding rules.
Example
- Class I recall, multi-channel distribution
- 1200 total clients from 5 distributors (400, 200, 300, 100 and 150 retailers) plus 50 retailers from the recalling firm's distribution list
- Sample size of 160 (from Table 5)
- Sampling interval = 1200/160 = 7.50 (leave two decimal points for calculating number of clients per distributor)
- The number of clients selected from each distribution list = the number of clients divided by the sampling interval with two decimal places
- At the recalling firm the number of clients to be selected is calculated to be 7 (50/7.50 = 6.66 which is rounded to 7)
| Recalling firm or distributors | Number of clients | Sampling interval (with decimals) | Number of clients divided by sampling interval | Number of clients to be selected |
|---|---|---|---|---|
| Recalling firm | 50 | 7.50 | 6.66 | 7 |
| Distributor 1 | 400 | 7.50 | 53.33 | 53 |
| Distributor 2 | 200 | 7.50 | 26.66 | 27 |
| Distributor 3 | 300 | 7.50 | 40 | 40 |
| Distributor 4 | 100 | 7.50 | 13.33 | 13 |
| Distributor 5 | 150 | 7.50 | 20 | 20 |
| Total | 1200 | 160 |
Data Entry:
- The lead investigator will record the number of clients to be selected on each distribution list in the IMS: "Recall tab, Eff. Check Summary tab, Number of Consignees tab".
Step 3: Select the clients for verification
3.1 Procedures to select clients
The lead investigator will immediately provide the assigned inspectors liaising with distributors with the recall verification plan, including the relevant IMS and recall numbers.
Using the recall verification information, the lead investigator and/or assigned inspectors will select the clients for verification using one of the following procedures:
- Sampling interval
- Random number generator
3.1.1 Sampling interval procedure
The lead investigator and/or assigned inspectors will use the lists of receiving clients and proceed as follows:
- Remove all duplicate entries, if any remain
- Randomize the order of the clients from each distribution list, if possible
- If the starting point has not been provided, randomly select a number between 1 and the sampling interval to determine the starting point
- Start at the top of the list and count down until the starting point is reached. This will be the first client selected for an effectiveness check
- Select every kth client, starting with the next client (where k, is the sampling interval)
When necessary, the lead investigator and/or assigned inspector will return to the start of the distribution list and continue selecting clients until the required number of clients is reached.
Example
- Class I recall, multi-channel distribution
- 5 distributors (400, 200, 300, 100 and 150 retailers) plus 50 retailers from the recalling firm's distribution list equals 1200 clients
- Sample size of 160 (see Table 5)
- Sampling interval of 7.50 (1200/160 = 7.50), rounded to 8 to identify the clients in the list where effectiveness checks will be carried out
- The number of clients selected from each distribution list = the number of clients divided by the sampling interval with two decimal places. At the recalling firm the number of clients to be selected is calculated to be 7 (50/7.50 = 6.66 which is rounded to 7)
- If the randomly selected starting point is 3, the first client selected from each distribution list will be the third client listed, followed by every eighth client selected thereafter until the desired number of clients has been selected
| Recalling firm or distributors | Number of clients to be selected | Procedure |
|---|---|---|
| Recalling firm | 7 | From the recalling firm's distribution list, the first client selected will be the 3rd client, and then every 8th client is selected thereafter. The selected clients will be 3, 11, 19, 27, 35, 43 and then 1 for a total of 7 clients. |
| Distributor 1 | 53 | For the first distributor (400 retailers), the selected clients will be 3, 11, 19, 27, 35, 43… 379, 387, 395, and then 4, 12, 20 for a total of 53 clients |
The same procedure for the selection of clients will be applied to the rest of the distributors.
3.1.2 Random number generator procedure
The lead investigator and/or assigned inspectors have the option to select the required number of clients by using a random number generator when the list of clients has not been randomized. This can be done by using random number generator software such as Research Randomizer. The lead investigator will use the distribution lists where all duplicates have been removed and will proceed as follows:
- Open a random number generator software, such as Research Randomizer
- Generate a list of random numbers: use one (1) as the lower range number and the total number of clients (in the specific list of clients) for the upper range number. Then, use the number of effectiveness checks required as the number to be generated (generate X random integers)
- After generating the desired random numbers, select the clients for verification. This is done by starting at the top of the list and counting down to the clients that correspond with the random number
Example
- Class I recall, multi-channel distribution
- 1200 total clients from 5 distributors (400, 200, 300, 100 and 150 retailers) plus 50 retailers from the recalling firm's distribution list
- Sample size of 160 (see Table 5)
- Sampling interval is 1200/160 = 7.50 (leave two decimal points for calculating the number of clients per distributor)
- The number of clients selected from each distribution list = the number of clients divided by the sampling interval with two decimal places (see section 2.6)
- At the recalling firm the number of clients to be selected is calculated to be 7 (50/7.50 = 6.66 which is rounded to 7)
- Using the number of clients on the distribution list and the number to be selected, the random number generator will create a list of random numbers which will then be used to identify the clients to be verified
| Recalling firm /distributors | Number of clients to be selected | Procedure |
|---|---|---|
| Recalling firm | 7 |
|
| Distributor 1 | 53 |
|
| For each distributor, please follow steps b) and c) above | ||
For each distributor, please follow steps b) and c) above
3.1.3 Selecting clients when the number of clients is less than the sampling interval
If the number of clients on a distribution list is less than the sampling interval, at least one client should be selected for verification from that distribution list.
Example
If the sampling interval is 14 and there are only 7 clients on a distribution list, then select at least one client for verification.
3.1.4 Creating lists of selected clients
The lead investigator and/or assigned inspectors may choose to create separate lists of selected clients or may choose to use the original distribution list with the selected clients clearly marked. Either method is acceptable as long as it is clear to the assigned inspectors conducting the verification activities which clients are to be verified.
3.1.5 Increasing or decreasing the number of clients for verification
Under exceptional circumstances, the number of clients to be verified may be increased or reduced.
Examples of exceptional circumstances may include
- Recalls involving a food distributed by clients that serve vulnerable populations such as the elderly, infants, pregnant women and the immunocompromised. In such circumstances, effectiveness checks should be conducted at all such clients (that is, 100% of these clients will be checked) which will result in an increase in the number of clients being verified.
- Recalls involving a food associated with a foodborne illness outbreak. In addition to the verification activities for the recall being given priority over verification activities for all other recalls, the approach may be to conduct additional effectiveness checks resulting in an increase in the number of clients verified.
- Recalls where the recalling firm or distributor conducts its own effectiveness checks immediately following the start of the recall and which have been deemed satisfactory by the CFIA. When the CFIA has sufficient confidence and evidence that the recalled food has been taken off the market, the number of clients being verified can be reduced.
- Recalls involving perishable food that is well past its best before date. Given that the food will not likely continue to be offered for sale or would be in an inedible state, the number of clients to be verified can be reduced
The method used to increase or reduce the number of clients for verification will be determined on a case by case basis. The lead investigator will consult with their supervisor and/or inspection manager, the area/regional recall coordinator and the OFSR to decide if the approach will be modified and if agreed upon, the appropriate approach to take.
Example
- Class I recall, multi-channel distribution
- 1200 clients (200 clients serve vulnerable populations and 1000 clients serve the general population)
The modified approach is to:
- Select 100% of the 200 clients that serve vulnerable populations for verification. The critical number would be zero (0) due to the nature of concern.
- Use a statistical approach to select clients from the list of 1000 clients that serve the general population. For a Class I recall with a multi-channel distribution and a list of 1000 clients, Table 4 indicates that the appropriate sampling table to use is Table 5: Selection of clients for Class I multi-channel. The sample size is 155 and the critical number is 7.
A total of 355 (200 + 155) clients will be verified. In contrast, if the approach had not been modified, a total of 160 clients would have been selected for verification.
If the modified approach results in a resource challenge such that all the required effectiveness checks cannot be completed in a timely manner, priority should be given to the effectiveness checks being conducted for clients that serve vulnerable populations.
When the decision is to modify the approach, the lead investigator will include a brief rationale in the effectiveness checks request, and any areas and regions assigned to conduct the verification activities will comply with this request.
The lead investigator will document both the decision as well as the modified method used in the IMS. Please refer to Appendix 2, Procedure 5: Documenting key decisions during food safety investigations and recalls for additional information.
3.2 Determine in which area(s) and region(s) each selected client is located
The lead investigator and/or assigned inspectors will determine in which area(s)/region(s) each selected client is located and clearly indicate this on the distribution list. Where it is noted that the area of distribution of the recalled food is different than previously indicated to the CFIA, the assigned inspector will inform the lead investigator, who will notify the OFSR.
Certain areas or regions maintain a list of the towns and districts that are located within their geographical area of responsibility. Where available, they can be referred to for greater specificity.
Once each of the clients has been assigned to an area, the lead investigator will determine the total number of clients to be verified by each area.
Data Entry:
- The lead investigator will record the number of clients to be selected in each Area in the IMS: "Recall tab, Eff. Check Summary tab, Number of Consignees tab, Atlantic, Quebec, Ontario and Western" fields. The IMS will automatically calculate the total number of clients to be checked.
Step 4: Prepare and send a recall effectiveness check request
4.1 Prepare the recall effectiveness check request
The lead investigator and/or assigned inspectors will include the following elements in their request:
- Relevant IMS and recall numbers
- Recall classification and depth
- Reason for the recall (for example, allergen, microbiological, extraneous matter, etc.)
- Recommended time frames for completion
- Any list(s) of selected clients
- Copy of the label(s) or sufficient food details
- Link to the recall determination report (if available)
- Copy of the recalling firm's notice of recall (where available)
- Copy of, or link to, the public warning (if applicable)
- Any other details which may help the assigned inspector(s) to complete the verification activities more effectively
Any subsequent distribution will be handled by the assigned inspectors using the same procedure, as stated above.
Please refer to Addendum 3: Recall effectiveness check request template.
If a recalled food-specific distribution list was not provided by the recalling firm or any distributors, the lead investigator will add a statement to the recall effectiveness check request advising that the distribution list provided was not food-specific and as such, some of the clients on the list may not have received the recalled food.
4.2 Provide a rationale when using a modified recall effectiveness check approach
If the recall effectiveness check approach was modified, the lead investigator will include a brief rationale in the recall effectiveness check request.
Example 1
This is a Class I recall to the consumer level with a public warning due to possible E. coli 0121 contamination of flour products. The recalled food is associated with a foodborne illness outbreak investigation. As such, verification activities are to begin immediately and to be given priority over verification activities for all other recalls (refer to section 3.1.5).
Example 2
This is a Class II recall to hotel/restaurant/institution level with a public warning due to possible Listeria monocytogenes contamination of ice cream sundae cups. The recalled food has been distributed to hospitals and senior residences, and after consultation, the verification approach is to be modified to increase the number of clients to be verified (refer to section 3.1.5).
4.3 Send the recall effectiveness check request to the appropriate area/regional recall coordinator
The lead investigator/assigned inspectors will send the recall effectiveness check request to the appropriate area/regional recall coordinators, copying their own area/regional recall coordinator. If there are clients in Quebec, the recall effectiveness check request will be sent to the Quebec effectiveness check recall coordinator and the Quebec area recall coordinator.
If the recall effectiveness check request is sent by the assigned inspectors in connection with a distributor's list, the lead investigator should also be copied on the request.
Data Entry:
- The lead investigator will record this, including a summary of the information sent and rationale for the request (if applicable), in the IMS: "Recall tab, Eff. Check Summary tab, Office Responsible tab". Enter the required information in the appropriate fields.
Step 5: Conduct recall effectiveness checks (assigned inspectors)
The assigned inspectors will conduct effectiveness checks with clients to determine if the clients have:
- been notified of the recall by their supplier (the recalling firm or distributor)
- taken the prescribed product action
The assigned inspectors will ensure that effectiveness checks are conducted in a timely manner so that the established time frames for completing the verification activities can be met (refer to Table 2). However, these time frames may need to be adjusted when the recall effectiveness checks are performed by other government authorities based on a Memorandum of Understanding and/or operational needs.
With copies of all supporting documents, including the label(s), the recalling firm's notice of recall and the CFIA's public warning (if applicable), the assigned inspectors will conduct the effectiveness checks according to the following provisions:
- For chain stores, effectiveness checks can be conducted via on-site visit or telephone call as either method is considered equally effective.
- For independent stores, 75% of the effectiveness checks will be conducted by on-site visit and 25% by telephone calls.
- In situations where the recall effectiveness check approach has been modified to increase the number of clients to be verified, on-site visits are preferred over telephone calls.
Please refer to Addendum 4: Recommended on-site and telephone verification procedure.
5.1 Gathering and recording information
The assigned inspectors will ensure the following information is gathered and recorded while conducting the recall effectiveness check:
- Name and address of the client (outlet)
- Method for conducting the recall effectiveness check (visit or telephone)
- Date the effectiveness check was conducted
- Name of the contact person at the client that is providing the information
- Whether notification advising of the recall was received by the client
- Whether uncorrected recalled food was available for sale at the time of the effectiveness check
- Quantity of uncorrected recalled food available for sale (if applicable)
- Name of the distributor who supplied the client (if applicable)
- Whether the client distributed the recalled food to other clients
- Whether the client repackaged, relabelled or reworked the recalled food and any relevant associated information
- A detailed description of the method used by the store to detain or dispose of the recalled food, and the amount disposed using this method
Inspectors may use the Effectiveness check worksheet as a template to assist in gathering the necessary information.
Where it is determined that the client further distributed the recalled food, the assigned inspectors will immediately request a detailed distribution list of their clients who received the recalled food (if known) and/or were notified of the recall.
Where it is determined that the client repackaged, relabelled or reworked the recalled food (for example, fresh cut cantaloupes were used to make fruit trays, deli meats or cheeses were used to make sandwiches or party trays, flour was used to make instore baked goods, etc.), a food safety investigation should be initiated immediately as the final food product may also pose a risk to human health.
Data Entry:
- The assigned inspectors will record the information in the IMS: "Recall tab, Eff. Check tab, Add", and enter the details of each recall effectiveness check.
5.2 Assistance conducting recall effectiveness checks
The effectiveness checks may be conducted by the CFIA or another government authority such as the ministère de l'Agriculture, des Pêcheries et de l'Alimentation du Québec. In such situations, the lead investigator, assigned inspector or the Quebec effectiveness check coordinator will follow up with the government authority to monitor the progress of the effectiveness checks.
In certain situations, the CFIA may request the assistance of local health units in conducting effectiveness checks. Assistance may be required when the recalled food is associated with a number of illnesses, there is an urgent need to remove it from the market quickly, it has a widespread distribution and/or is distributed at retail outlets, such as convenience stores, where the local health units have familiarity and jurisdiction. The lead investigator will consult with their supervisor and/or inspection manager, the area/regional recall coordinator and the OFSR to determine if the assistance of local health units will be requested with conducting effectiveness checks.
If assistance from a local health unit is required to conduct the effectiveness checks, please refer to Addendum 5 for additional guidance and templates.
5.3 Deviation assessments and actions
Deviations are occurrences where unfavorable results are encountered during the recall effectiveness check. When a deviation is identified the assigned inspector will:
- request immediate compliance by the client with the instructions provided by their supplier and/or in the notice of recall
- determine the reason for the deviation and, where appropriate, ensure corrective actions are initiated by the client
- classify the type of deviation (see section 5.3.1)
- inform the lead investigator, and if applicable the assigned inspector responsible for the distributor if the client is from a distributor's list, so the recalling firm and/or appropriate clients, are notified and requested to take appropriate action, where applicable
The assigned inspectors will continue with the effectiveness checks unless they are advised by the lead investigator to stop.
Note: If it is noted that recalled product is still for sale the assigned inspectors will oversee its' immediate removal. It must then be determined why appropriate product action was not taken. If the client did not receive a recall notice from the recalling firm, the lead investigator must be notified so follow-up at the recalling firm can be conducted.
5.3.1 Types of deviations
There are 2 types of deviations:
-
Product deviations
Product deviations are defined as those occurrences where the affected food has not been removed from sale or its label has not been corrected (see Table 1: Different scenarios regarding deviations). When entered into the IMS these are automatically identified as valid deviations. The assigned inspector is still required to enter a comment, explaining whether the deviation is valid or invalid, as it will be reviewed by the lead inspector when determining whether or not the recall was effective.
In such situations, the assigned inspector will ensure the food is immediately removed from sale and will take any necessary actions (refer to the Standard regulatory response process (accessible only on the Government of Canada network – RDIMS 9146498)). For more information regarding the action to be taken refer to Operational guidance: Assessing, monitoring and documenting the disposition of affected food products identified through food safety investigations, including recalled products (accessible only on the Government of Canada network – RDIMS 11350687).
Product Deviation Examples
- The receiving client received the notice of recall but the recalled product is still available to consumers.
- Affected product found on the store shelves has not been relabelled correctly.
- The receiving client received the notice of recall and removed the recalled product, however it was inadvertently put back on the shelf for consumers.
-
Process deviations
Process deviations are related to problems encountered during the implementation of the recall and do not count towards the calculated critical number (see Table 1: Different scenarios regarding deviations). However, the assigned inspector is still required to enter a comment about the deviation, as it will be reviewed by the lead inspector when determining whether or not the recall was effective.
Where process deviations are encountered, it may be concluded that the recalling firm or the receiving client did not properly follow its recall process or that the recall process may be ineffective. This holds true even if no product is found on the market and there is no ongoing consumer exposure, as process deviations were noted this portion of the recall process was ineffective. The assigned inspector and/or lead investigator, in consultation with their supervisor and/or the area/regional recall coordinator and the OFSR, will provide guidance to ensure the recalling firm/receiving client addresses the deviations.
Process Deviation Examples
- The notice of recall was not received by the receiving client, regardless of whether or not recalled product is found on the store shelves.
- A receiving client did not receive the notice of recall but became aware of the recall via an alternative method (for example, by the media).
Where it is found that a receiving client along the distribution chain was appropriately advised of the recall but did not notify the next level of distribution, the CFIA will take appropriate actions to ensure that the recall is properly conducted by all affected clients. The lead investigator will consult with their supervisor and/or area/regional recall coordinator and OFSR to determine whether or not it is necessary to ask the client (the distributor) to conduct the recall. Each situation will be considered on a case-by-case basis and will depend on factors such the reason for the process deviations, previous compliance history, etc.
| Notification received by client | Food found offered for sale | Type of deviation | Counts towards critical number |
|---|---|---|---|
| Yes | No | No deviation | No |
| Yes | Yes | Product deviation | Yes |
| No | Yes | Product deviation | Yes |
| No | No | Process deviation | No, but follow-up should occur to determine the reason for the deviation |
5.3.2 Deviation validity
Upon the identification of a deviation, the assigned inspector, in consultation with the lead investigator when needed, will determine if the deviation is due to a deficiency in the recalling firm's recall process. If the deviation was caused by the recalling firm, this is referred to as a valid deviation, and is taken into consideration by the lead investigator when determining whether or not the recall was effective. Following the identification of a valid deviation, the lead investigator must be notified to ensure appropriate follow-up and corrective actions are taken at the recalling firm.
Example
An example of a valid deviation is when a receiving client has been notified of the recall but the receiving client continues to offer the food for sale and the recalling firm did not verify that the requested action had been taken.
Where it is determined that the actions or non-actions of the recalling firm were not responsible for the identified deviation, this is referred to as an invalid deviation. Although some deficiency has been identified, it is not a direct result of the recalling firm's actions, but due to a deficiency in the distributor or receiving client's recall process. These deviations are to be recorded by the assigned inspector in the effectiveness check report in the IMS (refer to section 5.1) together with a comment indicating why the deviation occurred but are not taken into account by the lead investigator when determining whether or not the recall was effective. However, further follow-up and corrective actions must be taken at the distributor/client level.
Example
An example of an invalid deviation is when the receiving client advises the assigned inspector that they have not been notified of the recall, and upon follow up with the recalling firm (or distributor), the lead investigator (or assigned inspector for the distributor) is able to confirm through documentation that the receiving client was appropriately notified.
Determining whether the deficiency is a valid or invalid deviation allows for further focused investigation and implementation of corrective actions at the appropriate point in the distribution chain.
Step 6: Review of the recalling firm's activities (lead investigator)
For all classes of recall, the lead investigator will verify the effectiveness of the recall by conducting a review of the recalling firm's recall activities and their outcome. The lead investigator will review the following documents:
- The recall notices signed and returned by clients confirming action taken
- The logs/reports of notifications made to clients by telephone calls
- Documentation issued to clients for returned food
In situations where the recalling firm has a large number of receiving clients, the lead investigator uses his/her own best judgement, in consultation with their supervisor, to determine the appropriate number of documents to review.
During the review, the lead investigator will verify if the firm has documented key recall activity information such as the:
- date when the clients were contacted
- method of contact (for example, telephone, email, etc.)
- name of the person contacted at each client
- action taken by each client (for example, returned food to the firm's warehouse)
- quantity, and if readily available the lot code, of each recalled food available at each client's site at the time of the recall
When a deviation is identified the lead investigator will:
- request immediate compliance by the recalling firm
- determine the type of deviation (refer to section 5.3.1)
- determine the reason for the deviation and, where appropriate, ensure corrective actions are initiated by the firm
While conducting this review, if the lead investigator identifies concerns with a specific receiving client, they will initiate follow-up (or will notify the appropriate CFIA office to follow up) directly with the client in question to request compliance.
Step 7: Determine the overall effectiveness of the recall
The lead investigator will review the results of the effectiveness checks conducted by the assigned inspectors and the recalling firm's documentation relating to the recall. At this point the intention is to determine whether or not the recalling firm's recall process was effective.
During the evaluation of the deviations and associated reasons, the lead investigator determines if the valid deviations are:
- isolated
- systemic at the level of the recalling firm
- systemic beyond the recalling firm
Deviations which are isolated in nature have a limited effect on the overall recall process. However, if any of the deviations follow a pattern or trend it may be indicative of a widespread systemic problem. These types of deviations may have a more significant impact on the overall effectiveness of the recall process.
Examples
An example of an isolated reason for a product deviation is when the affected food was removed from sale at a single store but was re-shelved by mistake.
An example of systemic reason is when there is a breakdown in the notification process or a significant delay in the notification of all receiving clients by the recalling firm/distributor.
To assign an overall effectiveness rating to the recalling firm's recall activities the lead investigator will take into consideration both the number and the nature of all deviations:
- Does the number of valid product deviations exceed the calculated critical number?
- Are the validated product deviations isolated, systemic at the recalling firm or systemic beyond the recalling firm?
- What is the number and nature of the process deviations documented?
When the number of valid deviations exceeds the calculated critical number, having the recalling firm reissue the recall may be considered.
Even if the number of valid deviations is less than or equal to the calculated critical number in the sampling plan, there may be sufficient concern regarding the number and nature of the process deviations reported for the recall to be considered ineffective.
The lead investigator will classify the recall as effective or ineffective, which may be done in consultation with their supervisor and/or the area/regional recall coordinator (as per area protocol) when needed.
At any time during the recall verification process, if it is determined that a recall is ineffective, the lead investigator will:
- notify their supervisor, area/regional recall coordinator and the OFSR immediately to determine appropriate actions to be taken (for example, request that the recall is re-done by the recalling firm, issue a public warning, etc.)
- notify the recalling firm that the recall was found to be ineffective
- discuss the situation with the recalling firm to determine what action they will take to improve the effectiveness of the recall (for example, issuing additional recall communications, etc.)
For examples of effective and ineffective recalls, refer to Addendum 6.
The lead investigator will communicate their decision according to their established procedures. The decision should be documented in IMS as per Appendix 2, Procedure 5: Documenting key decisions during food safety investigations and recalls.
Data Entry:
- The lead investigator will record the recall rating and any comments in the IMS: "Recall tab, Eff. Check Summary tab, Conclusion tab, Recall Rating" field and "Comments on Recall Rating" field.
Step 8: Follow-up with the recalling firm
The lead investigator will discuss the overall effectiveness of the recall procedure with the recalling firm, identify any deviations that need to be addressed and/or request corrective actions as deemed appropriate.
Example
If the recalling firm was unable to provide the distribution list in a timely manner, the lead investigator will ensure that the recalling firm implements procedures to ensure that the list will be available in a timely manner.
Data Entry:
- If corrective actions are required, the lead investigator will summarize these actions in the IMS: "Recall tab, Eff. Check Summary tab, Conclusion tab, Corrective Action Summary" field.
If, after having been formally notified by the CFIA of the ineffectiveness of their recall, the recalling firm is unwilling or incapable of re-initiating or modifying its recall, the lead investigator, in consultation with their supervisor, inspection manager, area/regional recall coordinator and OFSR, will determine the appropriate actions to be taken by the CFIA.
Actions to be considered may include:
- public warnings issued by the CFIA
- food seizures
- initiating the mandatory recall process
- appropriate enforcement and/or compliance actions
| Recall classification | Verification activities will begin within __________ business Table Note a days of the initiation of a recall requested by the CFIA | Verification activities will be completed within __________ business Table Note b days of the initiation of a recall requested by the CFIA |
|---|---|---|
| Class I | 3 | 10 |
| Class II | 5 | 12 |
| Class III | 10 | 17 |
When effectiveness checks are conducted by another government authority, the time frames for completing the verification activities may need to be adjusted due to an existing Memorandum of Understanding and/or operational needs of the government authority.
| Step | Activity to be performed |
|---|---|
| 1 | Request a detailed distribution list of clients from the recalling firm
|
| 2 | Identify the appropriate recall verification plan to use:
|
| 3 | Select the clients for verification
|
| 4 | Prepare and send a recall effectiveness check request
|
| 5 | Conduct recall effectiveness checks (assigned inspectors)
|
| 6 | Review of the recalling firm's activities (lead investigator) |
| 7 | Determine the overall effectiveness of the recall |
| 8 | Follow-up with the recalling firm |
| Recall → distribution "X" |
Class I | Class II | Class III |
|---|---|---|---|
| Multi-channel | Table 5 at the prescribed rate | Table 6 at the prescribed rate | Table 7 at the prescribed rate |
| Single channel | Table 6 at the prescribed rate | Table 7 at the prescribed rate | Table 8 at the prescribed rate |
| Clients | Sample size | Critical number |
|---|---|---|
| > 10,000 | 200 | 9 |
| 5000 – 9999 | 195 | 9 |
| 3500 – 4999 | 190 | 9 |
| 2600 – 3499 | 180 | 8 |
| 2000 – 2599 | 175 | 8 |
| 1600 – 1999 | 170 | 8 |
| 1150 – 1599 | 160 | 7 |
| 1000 – 1149 | 155 | 7 |
| 850 – 999 | 150 | 7 |
| 750 – 849 | 145 | 6 |
| 650 – 749 | 135 | 6 |
| 550 – 649 | 130 | 6 |
| 500 – 549 | 120 | 5 |
| 400 – 499 | 115 | 5 |
| 350 – 399 | 110 | 4 |
| 290 – 349 | 95 | 4 |
| 255 – 289 | 85 | 3 |
| 230 – 254 | 80 | 3 |
| 175 – 229 | 75 | 3 |
| 150 – 174 | 65 | 2 |
| 125 – 149 | 60 | 2 |
| 100 – 124 | 50 | 1 |
| 90 – 99 | 45 | 1 |
| 75 – 89 | 35 | 1 |
| 65 – 74 | 30 | 0 |
| < 64 | 25 or 100% | 0 |
Note: Irrespective of the classification of the recall, corrective actions are to be taken, even if only one client is found to be selling the food. There is no tolerance for a client selling a recalled food.
| Clients | Sample size | Critical number |
|---|---|---|
| > 5000 | 110 | 5 |
| 3000 – 5000 | 105 | 5 |
| 2000 – 2999 | 100 | 4 |
| 1600 – 1999 | 95 | 4 |
| 1250 – 1599 | 90 | 4 |
| 1000 – 1249 | 87 | 4 |
| 850 – 999 | 84 | 3 |
| 750 – 849 | 80 | 3 |
| 650 – 749 | 77 | 3 |
| 550 – 649 | 74 | 3 |
| 500 – 549 | 70 | 3 |
| 400 – 499 | 69 | 3 |
| 350 – 399 | 65 | 2 |
| 300 – 349 | 61 | 2 |
| 250 – 299 | 58 | 2 |
| 225 – 249 | 55 | 2 |
| 200 – 224 | 52 | 2 |
| 175 – 199 | 50 | 2 |
| 150 – 174 | 45 | 1 |
| 125 – 149 | 40 | 1 |
| 115 – 124 | 35 | 1 |
| 100 – 114 | 32 | 1 |
| 80 – 99 | 32 | 1 |
| < 80 | 25 or 100% | 0 |
Note: Irrespective of the classification of the recall, corrective actions are to be taken, even if only one client is found to be selling the food. There is no tolerance for a client selling a recalled food.
| Clients | Sample size | Critical number |
|---|---|---|
| > 2000 | 70 | 3 |
| 1000 – 2000 | 65 | 3 |
| 750 – 999 | 55 | 2 |
| 600 – 749 | 50 | 2 |
| 400 – 599 | 45 | 1 |
| 350 – 399 | 40 | 1 |
| 325 – 349 | 35 | 1 |
| 100 – 324 | 30 | 1 |
| < 100 | 25 or 100% | 0 |
Note: Irrespective of the classification of the recall, corrective actions are to be taken, even if only one client is found to be selling the food. There is no tolerance for a client selling a recalled food.
| Clients | Sample size | Critical number |
|---|---|---|
| > 2000 | 50 | 2 |
| 1500 – 2000 | 45 | 2 |
| 1000 – 1499 | 40 | 1 |
| 750 – 999 | 35 | 1 |
| 500 – 749 | 30 | 1 |
| 400 – 499 | 28 | 1 |
| 300 – 399 | 26 | 1 |
| < 300 | 25 or 100% | 0 |
Note: Irrespective of the classification of the recall, corrective actions are to be taken, even if only one client is found to be selling the food. There is no tolerance for a client selling a recalled food.
Addendum 1: Recommended procedure for obtaining distribution list(s) of clients
Steps
The lead investigator and assigned inspectors liaising with distributors will:
- review recalling firm or distributor's history and file to understand the nature of their clients and their distribution system
- ensure that appropriate documents are available for reference (for example, a public warning, recall determination, label, etc.)
- prepare the questions to ask the recalling firm or distributor. Ensure that the questions avoid leading the respondent to answer with what they think the CFIA wants to hear
- call the recalling firm or distributor to request the distribution list of clients. If there is no or very limited collaboration, the lead investigator or assigned inspector will conduct an onsite visit to request the distribution list
- identify him or herself and ask to speak with the person that has the authority to respond to food recall activities and/or was involved in recall activities (for example, owner, manager, supervisor)
- explain the CFIA's verification procedure to the recalling firm or distributor – "The CFIA will be conducting a review of the results of [your/the recalling] firm's recall activities and verification of your clients' actions through the use of a statistical sampling plan"
Questions to consider
- "Please describe how your food is distributed including how orders are taken"
- "How does the food flow from your location to your clients?"
- "How do you track how the recalled food is sold?"
- "Is the food sold to clients only by the case or is the case broken?"
- "Do you know which of your clients received the recalled food?"
- If the firm is unable to provide a food-specific distribution list, ask:
- "Will you be notifying all of your clients and how?" or
- "How will you be narrowing the list down for notification?"
- If the firm is unable to provide a food-specific distribution list, ask:
- "Where is your food distributed?"
- "Can you clarify the geographical distribution by provinces?"
- "Do you export the affected food?" If yes, "To which countries?"
- "Do you distribute the affected food to cannabis licence holders?" If yes, "which licence holders?"
- "Do you sell directly to retailers?"
- "Do you sell to cash and carry outlets?"
- "Do you sell to distributors and fundraisers?" If yes, ask the firm to identify them.
- "Do any of your clients buy the recalled food to make their own food?" If yes, ask the firm to identify the manufacturers.
- "Was this food sold to clients who sell to vulnerable populations such as the elderly, infants, pregnant women and the immunocompromised?" If yes, ask the firm to identify these clients.
- "Was the food sold or otherwise provided to food banks?" If yes, ask the firm to identify them.
- "How quickly can you get the list of your clients to us?"
- "Approximately how many retailers sell the recalled food?"
- "How did you determine this number?"
Outcome
At the end of the discussion, the lead investigator and assigned inspectors (if there are distributors) will know:
- if the recalling firm or distributor can provide a detailed distribution list of clients within the time frames
- the actual number or best estimate of clients who are selling or could have received the recalled food
- area(s) and region(s) where the food was distributed
- if the recalled food was distributed to locations frequented by a vulnerable population
- if there are any distributors or manufacturers who bought the recalled food
Addendum 2: Determining whether the distribution is single channel or multi-channel
Accessible only on the Government of Canada network
Addendum 3: Recall effectiveness check request template
A. Request for recall effectiveness checks with distributors
This is a request for distribution lists.
Recall classification: (Enter the recall classification)
Depth of recall: (Enter the recall depth)
Reason for food recall: (Enter the hazard, manufacturer name, product name, product description: brand, size, lot code)
Recalling firm: (Enter name of recalling firm)
Please contact each of the distributors identified in your region on the attached distribution list to request a detailed distribution list of clients who received the recalled food.
Please also determine if each distributor you contact was notified of the recall, took appropriate product action as per the notice of recall issued by (enter name of recalling firm), and notified their clients of the recall. Please advise me (the lead investigator) of any clients that were not notified of the recall. Upon receiving any distribution list, please determine if there are any further distributors on the list and immediately notify the appropriate CFIA office to immediately contact those distributors to obtain their detailed distribution lists. Please notify me of any additional distributors.
Please remove duplicate and foreign clients, if any, on the list and advise me of the number of clients on each distribution list (excluding any distributors and fundraisers) you obtained. I should also be notified about foreign clients identified on the distribution list, so the Office of Food Safety and Recall can be informed and the respective foreign food authority notified.
Please record your findings in the Issues Management System number XXXXX, recall number XXXXX.
The recommended time frame for completion of verification activities for this recall is by Month, Day, Year.
The recall determination report can be found here [link, if available] and the public warning can be found here [link, if available].
Please find attached:
- The list of distributors.
- Copies of the label.
- Copy of the notice of recall.
B. Request for recall effectiveness checks with clients
Recall classification: (Enter the recall classification)
Depth of recall: (Enter the recall depth)
Reason for food recall: (Enter the hazard, manufacturer name, product name, product description: brand, size, lot code)
Recalling firm: (Enter name of recalling firm)
Please contact each of the selected clients identified in your region on the attached distribution list to determine if each client was notified of the recall by their supplier and have taken the prescribed product action.
Please advise me of any deviations.
If any of the clients turn out to be distributors, please notify the appropriate CFIA office to immediately contact the distributors to obtain their detailed distribution lists. Please notify me (the lead investigator) of any additional distributors.
If you identify any clients that repackage, relabel or rework the recalled food, please immediately initiate a food safety investigation and advise me.
If you identify any foreign clients, please inform me immediately to ensure the Office of Food Safety and Recall is informed and the respective foreign food authority may be notified.
Please record your findings in the Issues Management System number XXXXX, recall number XXXXX.
The recommended time frame for completion of verification activities for this recall is by Month, Day, Year.
The recall determination report can be found here [link, if available] and the public warning can be found here [link, if available].
Please find attached:
- The list of clients.
- Copies of the label.
- Copy of the notice of recall.
Addendum 4: Recommended on-site and telephone verification procedure
The lead investigator and assigned inspectors will:
- ensure that appropriate documents are available for reference (for example, the public warning, recall determination report, the label, etc.)
- have a blank effectiveness check worksheet form. Print one from CFIA's forms catalogue #5128
- prepare the questions to ask the client. Ensure that the questions avoid leading the respondent to answer with what they think the CFIA wants to hear
- call or visit the client. If a visit is conducted, check the store shelves to see if the food is being offered for sale
- identify him or herself and ask to speak with the person that has the authority to respond to food recall activities and/or was involved in recall activities, for examples, owner, manager, supervisor
- explain the general purpose of your call or visit – "A food recall has been initiated. The CFIA is conducting verification activities to determine the effectiveness of the recalling firm's recall efforts. Your [store] was included in the distribution list provided to us by the recalling firm"
Questions to consider
- "Please tell me about the recall notice?" Let the person describe the situation and then, follow-up with specific questions, as necessary:
- "Are you aware of a food recall conducted by "X" of food "Y"?"
- "When did you become aware of this recall? Who notified you and how did they notify you?" If the client was notified by fax or letter, ask to review the notice and verify the date of receipt.
- "What happened at the store once the notice was received? Let the person describe the activities and follow-up with the specific questions, as necessary:
- "What quantity of the recalled food was on your store shelves and your storage area/stockroom at the time the notice was received from the "X"?"
- "What action was taken on this food?"
- "What measures are taken to ensure the recalled and segregated food is not mistakenly returned to store shelves?"
- If food disposition has taken place, obtain a detailed description of the disposition method used (for example, was the food returned to the recalling firm? Was the food thrown away and how? Was the food relabelled?)
- If disposition has not taken place, physically verify the disposition of the food, if on-site
- "Do you (and how will you) inform your head office /manager/owner once the food has been removed / disposed?"
- "Other than retail sales to individual consumers, do you further distribute this food (act as a wholesaler)?
- If so, have you taken any measures to relay the recall information to your clients?
- When, how and by whom was this done? If the client was notified by fax or letter, ask to see a copy of the notice.
- "The CFIA will need to follow up with these clients as well, when could we receive a copy of your distribution list"
- "Do you repackage, relabel, further process or rework the recalled food?" If yes, initiate a food safety investigation.
Outcome
At the end of the effectiveness check, the lead investigator and assigned inspectors should make sure that they have answers to all the questions on the effectiveness check worksheet form. If there has been a deviation, the lead investigator and assigned inspectors should request immediate compliance.
Addendum 5: Requesting assistance from local health units with conducting recall effectiveness checks
If assistance from local health units is required to conduct the effectiveness checks, the following templates have been provided as examples and may be used or modified as needed:
- Letter to local health units for requesting assistance with conducting recall effectiveness checks
- Instructions for completing effectiveness checks for food recall recording form
- Effectiveness checks for food recall recording form
- Recall effectiveness check summary form
Addendum 6: Examples of effective and ineffective recalls
The lead investigator will assign the effective or ineffective classification to the recall.
Using the example of 1200 clients for a class I recall, with verification done at 160 randomly selected clients and a critical number of 7:
-
All clients that were checked were found to have received the notice of recall from the recalling firm and to have removed the food from sale.
Action: None. The recall is effective.
-
Eight clients that were checked were found to have deviations. Four clients have not received the notice of recall and were still offering the food for sale. The other four clients have received the notice, but they have not taken the requested food action. Therefore, the food remains available to the consumer at eight clients, exceeding the critical number.
Action: The recall is deemed ineffective as there are eight product deviations, in other words, a total of eight clients still had the food available for sale, which is one more than the critical number of seven. Product action needs to be taken and the recall may need to be repeated by the recalling firm. Further investigation is required to determine why the clients who received the information did not take the appropriate action. The inspector will ensure appropriate corrective actions are put in place and implemented to prevent similar incidents in the future.
-
Ten clients that were checked were found to have deviations. Seven were found to have process deviations and three were found to have product deviations.
Action: Overall, the recall appears to be effective as the three product deviations are less than the critical number of seven. However, the recalling firm may not have an appropriate recall plan or process.
The recall may be considered ineffective if the reasons for the deviations were due to widespread systemic issues. The recall may be considered effective if the reasons for the deviations are isolated in nature.
The CFIA will investigate to determine the root cause of the deviations, ensure that the recalling firm and/or the affected clients address the deviations immediately and will provide guidance, as required. Product action will be taken and the recall may need to be repeated by the recalling firm. Further investigation is required to determine why the clients who received the information did not take the appropriate action. The inspector will ensure appropriate corrective actions are put in place and implemented to prevent similar incidents in the future.
-
Six clients that were checked were found to have process deviations. None of these clients received the notice of recall; however the product was no longer available for sale.
Action: Overall, the recall appears to be effective as no product was found to be for sale. However, the recalling firm may not have an appropriate recall plan or process. The recall may be considered ineffective if the reasons for the deviations were due to an issue at the recalling firm. The CFIA will take appropriate measures to ensure that the recalling firm and/or the affected clients address the deviations immediately and will provide guidance, as required.
Procedure 2: Best practices for storing food safety investigation information
Accessible only on the Government of Canada network.
Procedure 3: Tampering
1.0 References
- Operational Procedure: Enforcement and Investigation File Referral Standard (EIFRS) (accessible only on the Government of Canada network)
- Enforcement and Investigation File Referral Standard (EIFRS) checklist (accessible only on the Government of Canada network)
- Compliance and Enforcement Policy
- Standard regulatory response process
- Food regulatory response guideline
- Operational Procedure: Procedure for seizure and detention, authorizing movement and disposition
- Operational Procedure: Issuing a Letter of Non-Compliance
- Operational Procedure: Conducting a meeting with the regulated party
2.0 Introduction
According to the Safe Food for Canadians Act, "it is prohibited for a person to tamper with any food commodity, its label or its package with intent to render the food commodity injurious to human health or cause a reasonable apprehension in others that the food commodity is injurious to human health. It is [also] prohibited for a person to threaten to render a food commodity injurious to human health".
Tampering with food is a punishable offence under the Criminal Code of Canada.
A person may tamper with food for a variety of motives, including:
- extortion
- for the personal gain of the perpetrators
- promotion of political objectives
- drawing attention to a cause
- revenge (for example, by causing financial damage to a regulated party through lost sales)
- mischief or prank
- terrorism
- reasons known only to the perpetrator
3.0 How to determine whether an incident is a suspected tampering incident
The following are examples of questions to consider to determine whether an incident should be treated as a suspected tampering incident:
- Is there visible evidence that the integrity of the package has been compromised (for example, tears, perforations, loss of vacuum, open or broken seal)?
- Does the food appear to have been altered or replaced?
- Was extraneous material of a tampering nature found in the package or food?
- Is it possible that the orientation of the extraneous material in the package indicates whether the extraneous material was introduced deliberately or accidentally?
- Could the extraneous material (type, dimensions, condition) make it through the manufacturing process intact?
- Does the extraneous material seem consistent with the food manufacturing process (for example, if a screw was found, try to attribute its loss to the machinery)?
- Have other similar complaints been received by the regulated party?
- Has anyone indicated that the food has been tampered with or threatened to tamper the food, including any post on social media?
- Does the source of information and related details appear credible?
- Are there similar complaints in the Issues Management System (IMS)? Do the complaints appear to be related?
- Is the complainant an employee, former employee or competitor of the regulated party?
- Has there been recent labour unrest, disgruntled employees, layoffs or dismissals at the regulated party?
- Has the regulated party been associated with a previous tampering incident?
- Does the food look or smell any different than normal?
4.0 Roles and responsibilities
4.1 The police
As tampering is a criminal act, the primary responsibility for the investigation rests with the police. Local police conduct investigations at the local level while the provincial police and the Royal Canadian Mounted Police conduct investigations that have province-wide or national implications.
4.2 The Canadian Food Inspection Agency (CFIA)
The CFIA plays an important role in the initial identification of tampering incidents and will work with the police authority in charge in the ultimate resolution of the incident from a food safety perspective.
In situations where the regulated party has established emergency response and/or tampering protocols, the CFIA will work with them to coordinate the overall response to address public health and security concerns.
5.0 Tampering procedure
5.1 The Area
The Area will assign the highest priority to the tampering investigation and will give priority to any reports of atypical injury or illness directly attributable to a food.
The Area (either the inspector, supervisor or inspection manager) that initially receives the information of a suspected or confirmed tampering incident will immediately inform:
- the Area/Regional Recall Coordinator (ARC/RRC) where the food originated
- the Office of Food Safety and Recall (OFSR)
- the Manager, Area Enforcement and Investigation Services (EIS), who will contact the appropriate law enforcement agencies, such as local police or the Royal Canadian Mounted Police
- the regulated party responsible for the affected food
- any other parties within the CFIA, as appropriate
The director generals and directors of operations will establish a liaison network with regional hospitals, police authorities and related public health partners to ensure prompt sharing of information and investigation of tampering incidents.
The Area will follow their own tampering protocol based on the Incident Command System, which clearly delineates the roles and responsibilities of each participant as well as the resources, expertise and laboratory capabilities available to assist in the tampering investigation.
In situations where a recall is requested, the lead investigator will remain the main contact with the regulated party as per section 5.1.2.
5.2 The OFSR
In the event of a reported case of confirmed or potential food tampering or threat, the OFSR will coordinate the emergency response in collaboration with the Area.
The OFSR will notify:
- the National Issues Management Office
- the Executive Director, Inspection Support Directorate
- the EIS Manager
- Communications and Public Affairs Branch, as appropriate
- international food regulatory agencies, as appropriate
- key industry associations, as appropriate (for example, when a consumer advisory is issued)
The OFSR may request a health risk assessment, as per section 3.2. As a result of the health risk assessment, a voluntary food recall may be requested and a food recall warning may be issued to ensure the effective and efficient removal of food from the marketplace.
The OFSR may issue a consumer advisory when the determination of the validity of tampering allegations is not possible, unavailable or incomplete.
In the circumstances above, the CFIA will encourage the regulated party to issue a general public warning through traditional and social media, either nationally or locally, as appropriate; and through specialized news media (for example, professional or trade press, or to specific segments of the population, such as physicians, hospitals, pharmacists, etc.).
5.3 Food Safety Science Directorate
The Executive Director, Food Safety Science Directorate will coordinate the national laboratory capability and capacity when the tampering incident hazard is unknown, unusual or requires specialized analysis.
5.4 Area Enforcement and Investigative Services (EIS)
EIS will take the lead on facilitating coordination with the appropriate law enforcement agencies, such as local police or the Royal Canadian Mounted Police. EIS is also responsible for taking enforcement actions for infractions of the Safe Food for Canadians Act.
The Area EIS Manager should be included in the early stages of all tampering incidents. EIS may provide assistance regarding seizure, detention or handling of any suspect food, as it may be evidence of a criminal offence. The EIS Manager will also assess the adequate timing for an EIS investigation to be initiated.
Procedure 4: Requesting information from regulated parties during food safety investigations and recalls
This guidance applies to all food safety investigations and recalls whether handled within the normal scope of operations or when the Incident Command System has been activated.
1.0 Responsibilities
Lead investigator
During a food safety investigation, the lead investigator:
- coordinates and prioritizes the requests for information; and
- acts as the single point of contact with the regulated party in question.
If due to operational needs an alternate inspector, who is not the lead investigator, is assigned to take the liaison role with the regulated party, the Alternate will act as the single point of contact through whom all requests for information to the regulated party will be made. In such cases, the Alternate will not assume the remaining responsibilities of the lead investigator.
Inspector
The inspector who is part of the food safety investigation team assists in food safety investigations by collecting information and verifying accuracy and completeness of the facts while at the regulated party's facility. When the required information is not available or cannot be provided immediately by the regulated party the inspector takes note of the missing information and informs the lead investigator.
Operations branch management
Operations branch management provides guidance to the lead investigator on the prioritization of requests and supports the role of the lead investigator as the single point of contact for the Canadian Food Inspection Agency (CFIA). During normal operations, Operations branch management includes supervisors, inspection managers and executive level management; when using an Incident Command System, this includes the Incident Commander and the Operations Section Chief.
2.0 Procedure
The following provides guidance on how to request information during food safety investigations from regulated parties or their representative.
Establishing the line of communication
The lead investigator and where applicable, the alternate inspector, will be identified early in the food safety investigation and this information will be communicated within the CFIA and with the regulated party. This is particularly important when more than one inspector is involved (for example, for food safety investigations managed by a team; in establishments where there is daily CFIA presence; when the regulated party's head office is in a different location from the implicated manufacturing facility; and when an Incident Command System has been activated).
All requests from the CFIA to the regulated party or their representative will be conveyed through the lead investigator, or where applicable, the alternate inspector.
When first contact is made with the regulated party, the lead investigator, or where applicable, the alternate inspector will:
- explain his/her role in the food safety investigation and as the single point of contact for the CFIA;
- ask the regulated party to identify the appropriate contact, and an alternate, in their organization to whom requests related to the food safety investigation should be submitted; and
- confirm with the regulated party the best method for timely written communication, such as e-mail, fax, etc.
When deemed necessary, a meeting with representatives of the regulated party will be organized by the CFIA at the onset of the food safety investigation to explain the process and clarify roles and responsibilities. For more complex situations, or upon request from the regulated party, Operations Branch Management may be engaged and lead the meeting. Such meetings will include the lead investigator.
If due to operational needs the lead investigator or the alternate inspector changes during the course of the investigation, the inspector who is the current CFIA contact will notify the regulated party of the change and will provide the new inspector's details in writing. The new inspector will be the single point of contact for the CFIA.
Requesting information
During a food safety investigation, the regulated party will assist inspectors by providing the required information to them while they are on-site. When the required information is not available or cannot be provided immediately by the regulated party, inspectors will take note of the missing information and will inform the lead investigator for the purpose of coordinating requests.
The lead investigator will coordinate and convey any request to the designated regulated party contact, preferably in writing (by e-mail or other means as previously agreed). The request will clearly outline the required information and the expected timeline for providing it.
If the request is made verbally, the lead investigator will document it in his/her notebook and in the Issues Management System (IMS).
The following requests must always be made in writing:
- Complex requests (for example, there are many elements in the request)
- In situations where the regulated party has not actioned in a timely manner a request that was made verbally
- In the event of a recall, the receiving client list specific to the affected food and any missing affected food labels
When necessary and appropriate, the request will prioritize the various elements of information required (for example, when there are many elements required which do not have equal priority; when requests for information are coming from multiple groups within the CFIA). Lead investigators will consult with their supervisor, as necessary, for advice on prioritizing the elements of the request.
To ensure that the regulated party fully understands any written request, the lead investigator will maintain regular verbal dialogue with the regulated party contact.
Operations branch management or members of an emergency response team may wish to have a dialogue with the regulated party for complex situations or to reinforce the requests made by the lead investigator. In such cases, the lead investigator will be included in the dialogue.
Procedure 5: Documenting key decisions during food safety investigations and recalls
Accessible only on the Government of Canada network.
Procedure 6: Expediting the issuance of a public warning
1.0 Purpose
To provide guidance on the circumstances under which the issuing of a Food Recall Warning is to be expedited and on the associated process.
2.0 Scope
This procedure applies to certain food safety investigations and recalls where it is anticipated that the risk mitigation decision will include the issuance of a Food Recall Warning.
3.0 Background
This process includes sharing pertinent information from the draft Food Recall Warning with the implicated firm for its review for accuracy before the Canadian Food Inspection Agency (CFIA) risk mitigation decision has been made.
In most cases, successful collaboration with the firm in this process has allowed the issuing of Food Recall Warnings with the company being identified as taking action. In other instances, when the company was not reachable or did not agree to conduct a recall, the CFIA has issued Food Safety Warnings where the CFIA advised the public not to consume the affected food. Although the process has resulted in the issuance of public warnings in a timely manner, the CFIA is looking at ways to further gain efficiency wherever possible, particularly for incidents of highest public health importance.
The CFIA has put in place this procedure to be used for certain situations when, based on preliminary risk analysis, the CFIA has sufficient confidence that a public warning will be necessary and that it would need to be issued the same day. In these situations, the CFIA may share the pertinent information from a draft Food Recall Warning, accompanied by a disclaimer statement, with the implicated firm either before a health risk assessment is obtained and/or before the final CFIA risk mitigation decision is made. The disclaimer statement will indicate that the procedure for sharing information from the draft Food Recall Warning before a risk mitigation decision is made does not constitute a formal request by the CFIA to the company to conduct a voluntary recall of the food in question.
It is anticipated that the application of this procedure will further gain efficiency for the issuance of some public warnings.
4.0 Responsibilities
Roles and responsibilities for food safety investigations and recalls are described in this manual and the Food Safety Emergency Response Functional Plan.
Vice-President, Operations Branch
The Vice-President, Operations Branch or delegate is responsible for determining when there is sufficient confidence that the anticipated risk mitigation measure will include a public warning and when this procedure should be applied by CFIA Operations.
Executive Director, Inspection Support Directorate
The Executive Director, Inspection Support Directorate may make the decision on behalf of the Vice-President of Operations for those incidents that have clearly defined parameters in this procedure. This responsibility may be delegated to the Director, Office of Food Safety and Recall (OFSR).
OFSR Staff
The OFSR staff are responsible for:
- sending via email the pertinent information from the draft Food Recall Warning and a disclaimer statement to the lead investigator for sharing with the regulated party before the final CFIA risk mitigation decision is made;
- issuing a Food Safety Warning in the event that the company:
- does not confirm the accuracy of the information within the two hour time frame allotted;
- is not reachable within 2 hours of the lead investigator initiating contact, after all reasonable attempts by telephone and email have been made; or
- refuses to conduct the recall without delay and the level of risk is such that risk communication cannot wait; and
- approving the French and English versions of the Food Recall Warning and posting them on the Recalls and safety alerts website.
Lead investigator
The lead investigator (or the inspector assigned to take the liaison role with the regulated party) is responsible for:
- reviewing the email with information from the draft Food Recall Warning for accuracy;
- sharing the information and a disclaimer statement with the regulated party for their review of its accuracy, when requested by the OFSR;
- recording in the Issues Management System (IMS) the date and time when:
- the information is shared with the regulated party
- the regulated party confirms the accuracy of the information
- notifying the regulated party when the health risk assessment and/or risk mitigation decision is available; and
- obtaining the regulated party's agreement to proceed with a voluntary recall, if necessary. The date and time when the regulated party's decision is received is to be recorded in the IMS.
5.0 Procedure
This procedure is to be followed by CFIA Operations when authorized by the Vice-President of Operations or his delegate.
The Vice-President of Operations or his delegate determines when preliminary risk analysis provides sufficient confidence that the anticipated risk mitigation measure will include issuing a public warning, on the same day, for the following incidents:
- Any incident involving baby/infant food
- Listeria monocytogenes in a ready-to-eat food where there has been reported illness
- E. coli O157:H7 in ground beef where there has been reported illness
- Any issue of tampering which involves:
- A baby/infant food
- Multiple occurrences of the same nature
- When a National Incident Command System has been activated
- Any other incident at the discretion of the Vice-President, Operations Branch
In these circumstances, an email with the pertinent information from the draft Food Recall Warning is shared with the regulated party, before the final CFIA risk mitigation decision is made (in other words, either while the health risk assessment is being undertaken by Health Canada, when applicable, or at any time the Vice-President, Operations Branch, or delegate, has sufficient confidence that the anticipated risk mitigation measure will include a public warning).
The email is prepared by the OFSR and is shared with the regulated party through the lead investigator:
"The CFIA has requested Health Canada to conduct a health risk assessment on your food. The health risk assessment is a scientifically based process which determines the level of risk your food presents to the Canadian population. Once the health risk assessment is received, the CFIA will make a risk mitigation decision as to whether or not a recall will be requested and/or if a Food Recall Warning will be issued.
Based on the nature of this food safety incident and potential need for a timely response, the CFIA has prepared a draft Food Recall Warning to be used in the event that it is required as a risk mitigation measure. Pertinent information from the draft is being shared with you now so that you can review it for accuracy with respect to the food description, distribution and company name. Your confirmation regarding the accuracy of the information will expedite the issuance of a Food Recall Warning should it be required.
Your confirmation is required within the next two hours. Please note that, at this time, this does not constitute a request for your company to conduct a voluntary recall of the food in question. Furthermore, your confirmation regarding the accuracy of the information does not imply that you agree that your food poses a risk. The risk level will be determined by the health risk assessment.
The CFIA will notify you whether or not risk mitigation actions are needed as soon as the health risk assessment is received. In the event that a Food Recall Warning is required, it will be sent for distribution to the media and for publication on the Recall and safety alerts website shortly after the risk mitigation decision has been made, providing your confirmation of its accuracy has been received. Should we not receive your confirmation by the time the CFIA is ready to issue the warning (where required) and the two hours allocated for review have elapsed, the text of the first line of the release will be amended to reflect that the CFIA is issuing the warning."
The lead investigator records the date and time when the information from the draft Food Recall Warning was shared with the regulated party in the IMS. The two hour review period commences from this time.
When the information and disclaimer statement is provided by e-mail, the lead investigator immediately follows up with the regulated party by telephone to confirm that the email was received.
The lead investigator records the date and time when the regulated party confirms the accuracy of the information from the draft Food Recall Warning in the IMS and informs the OFSR of the regulated party's approval.
The lead investigator communicates the health risk assessment and/or risk mitigation decision when available to the regulated party. If this involves a request for the regulated party to conduct a voluntary recall, the lead investigator seeks a confirmation of their agreement that they will proceed with the recall.
In the event that the risk mitigation decision involves issuing a Food Recall Warning and the regulated party has not yet confirmed the accuracy of the draft, the lead investigator verifies the status of the review with the regulated party. In the event that the risk mitigation decision was made prior to the two-hour review period elapsing, the Food Recall Warning will not be issued until either the regulated party responds to the review request or the two-hour timeframe is complete.
Once the regulated party has confirmed the accuracy of the information and, when applicable, has agreed to conduct the recall, the OFSR approves the French and English versions of the Food Recall Warning and posts them on the Recalls and safety alerts website.
In the event that the regulated party does not confirm the accuracy of the information or is not reachable by the time the risk mitigation decision has been made and the two hours allotted for review have elapsed, or, when applicable, does not agree to conduct a recall, the OFSR will consider issuing a Food Safety Warning.
Where a regulated party does not agree to conduct a voluntary recall (when applicable), other actions, which are described in Section 5, may be considered by the CFIA.
Procedure 7: Responding to a company's request for a copy of a risk assessment
1.0 Purpose
To outline the process for responding to a request from a regulated party for a copy of a risk assessment, while ensuring personal and third party confidential business information is protected.
2.0 Responsibilities
Regulated party
Regulated parties can request a written copy of any risk assessment conducted on their food.
Lead investigator
The lead investigator (or the assigned inspector acting in the liaison role with the regulated party) is responsible for:
- notifying the Office of Food Safety and Recall (OFSR) of the request
- recording, in the Issues Management System (IMS), the date and time when:
- the request for a copy of a risk assessment is received from a regulated party
- the PDF copy of the assessment is shared with the requestor
Food safety recall specialist, Food Safety Investigation and Recall, OFSR
The food safety recall specialist is responsible for coordinating the OFSR's response to the request for a copy of a risk assessment.
Technical risk assessor, Food Safety Technical Assessment and Analysis, OFSR
The technical risk assessor is responsible for retrieving the risk assessment in question and ensuring that any protected information is redacted prior to its release. When the risk assessment originates from Health Canada, the technical risk assessor is responsible for informing Health Canada of the request and providing them with a redacted copy of the released document.
3.0 Procedure
A regulated party responsible for a food which was subject to a risk assessment may request, either verbally or in writing, a copy of the risk assessment.
After receiving a request for a copy of a risk assessment from a regulated party, the following steps will be followed:
- The lead investigator promptly sends an e-mail with the request to the food safety recall specialist assigned to the file and documents the request in the IMS.
- The Food Safety Recall Specialist:
- acknowledges receipt of the request and advises the lead investigator to document the request in the IMS
- determines if the requester is the recalling firmFootnote 10
- if the requester is the recalling firm, the Food Safety Recall Specialist will inform the lead investigator that a copy of the risk assessment can be shared and will be forwarded as soon as all steps of the Canadian Food Inspection Agency (CFIA)'s procedure for its release have been undertaken
- if the requester is not the recalling firm, the Food Safety Recall Specialist will inform the lead investigator that a copy of the risk assessment cannot be shared and will send the lead investigator the template message, found in Addendum B, to use when communicating this to the requester
- informs the assigned technical risk assessor of the request by e-mail, copying the National managers of Food Safety Investigations and Recalls and Food Safety Technical Assessment and Analysis, if the requester is the recalling firm
- The technical risk assessor:
- informs Health Canada of the request, when the risk assessment originated from Health Canada
- reviews the risk assessment for the presence of any personal information or confidential business information relating to a third party; any such information is redacted. If there is any doubt whether information should be protected, advice is to be sought from Legal Services and/or the Access to Information and Privacy Office, as appropriate
- creates a PDF copy of the risk assessment with the protected information redacted; and
- e-mails the final version of the PDF to the Food Safety Recall Specialist, copying the National managers of Food Safety Investigations and Recalls and Food Safety Technical Assessment and Analysis
- The title of the e-mail should state, "IMS xxxxxx - Copy of (Health) Risk Assessment as requested"
- The body of the e-mail should make specific reference to the request, for example, "Further to the request for a copy of the (Health) Risk Assessment made by…(identity of the requestor) on….(date)"
- The Food Safety Investigation and Recall Specialist:
- reviews the PDF file to ensure that all confidential business information and personal information has been redacted
- requests the technical risk assessor to make necessary changes as appropriate
- emails the final document with the heading "IMS xxxxxx - Copy of (Health) Risk Assessment as requested" to the lead investigator, copying the technical risk assessor. The text in the e-mail should make specific reference to the request, for example, "Further to the request for a copy of the (Health) Risk Assessment made by…(identity of the requestor) on….(date)". The e-mail should also advise the lead investigator to document the date and time when the risk assessment is released to the requester and that a conference call can be set up with all parties should the requester have any questions
- documents in IMS the date when the risk assessment was sent to the lead investigator
- The technical risk assessor sends the final PDF copy to Health Canada when the risk assessment originates from Health Canada.
- The lead investigator:
- forwards the PDF of the risk assessment to the regulated party. The accompanying e-mail, letter or fax should make specific reference to the request, for example, "Further to the request for a copy of the (Health) Risk Assessment made by… (identity of the requestor) on…(date)". )". The e-mail should also advise the regulated party that a conference call can be set up with all parties should they have any questions
- documents in the IMS the date when the risk assessment was sent to the regulated party
In the event that the regulated party has questions about the risk assessment, a conference call should be set up at a mutually convenient time for all parties involved, including Health Canada, when the risk assessment originated from them.
Addendum A: Types of information that must be protected
Personal information
Personal information refers to information about an identifiable individual that is recorded in any form (refer to the Privacy Act, Section 3, for the detailed interpretation of what constitutes "personal information"). In the context of a food safety investigation, the CFIA inspects and collects a wide variety of information which may include personal information on regulated parties' employees, suppliers/buyers' employees, and other complainants.
Personal Information that must be protected includes, but is not limited to:
- name, address, telephone number, e-mail address and websites (only if it discloses personal information about an identifiable individual);
- race, national or ethnic origin, religion, age and marital status;
- educational, medical, criminal or employment history;
- any identifying number or symbol assigned to the individual;
- personal opinions or views; and
- financial information.
Note: Information about an employee of a federal institution that relates to his/her position or functions is not personal information. Such information includes, but is not limited to:
- name, title, business address and telephone number;
- responsibilities of the position held by the individual; and
- personal opinions or views given in the course of employment.
Confidential Business Information
According to the Access to Information Act, confidential business information includes financial, commercial, scientific and technical information supplied to a government institution by an outside party that is treated consistently in a confidential manner by the outside party. In the context of a safety investigation, the CFIA inspects and collects a wide variety of information which may include potential confidential business information.
Examples of confidential business information that must be protected include:
- product formulations or recipes;
- scientific or technical information, such as unique manufacturing processes, research and development information;
- details on equipment and processes used in the plant;
- commercial information, such as sales, volume of production, food distribution and buyers and supplier information; and
- financial information.
Note: Business Information such as the name of the company may not be protected, please contact the Access to Information and Privacy Office for a decision to disclose this information.
Addendum B: Recommended messaging for co-packed food
RE: Request for a copy of the (Health) Risk Assessment regarding (Subject)
This is further to your request dated (date) for a copy of the (health) risk assessment regarding (subject).
Although the food in question carries your brand name, much of the information contained in the risk assessment relates to a third party (the recalling firm). For this reason, the release of copies of (health) risk assessments outside the formal Access to Information Process is done only for companies identified as recalling firms. However, the recalling firm (or your supplier) may request a copy which they may be willing to share with you.
If you wish to obtain a copy of the health risk assessment from the CFIA, you may make an Access to Information Act request online via the CFIA's website or in writing at:
Access to Information and Privacy Office
Canadian Food Inspection Agency
1400 Merivale Road
Ottawa, ON
K1A 0Y9
The request requires the payment of a $5.00 application fee. The Access to Information and Privacy Office will review your request and determine if any exemptions apply.
You can contact the Access to Information and Privacy Office by phone at (613) 773-5990 or by email at ATIP-CFIA-AIPRP@inspection.gc.ca for any information on this process.
You can also find general information on the CFIA website.
Please do not hesitate to contact me if you have any further questions.
Sincerely,
(lead investigator or assigned inspector)
Procedure 8: CFIA guidelines for early public communication of food safety incidents
Issue
The Canadian Food Inspection Agency (CFIA) has developed the following guidelines for determining when early public communication may be necessary to respond to food tampering situations or to address concerns raised by the public regarding food safety incidents. In these instances, public communication may be warranted prior to establishing a health risk and determining whether a food recall is necessary.
Background
The Health Portfolio department and agencies issue messages to Canadians that are general in nature in respect of food safety. In such instances, CFIA's Communications and Public Affairs is the lead and will work with appropriate portfolio partners.
Another well-established process for the CFIA is to issue Food Recall Warnings or Food Safety Warnings as risk mitigating measures when specific food has been assessed as representing a health risk. Such public communications typically occur after information on an incident is collected and verified through a food safety investigation and a health risk assessment is provided by Health Canada, or when a company recalls food on their own initiative. In 2014, the CFIA developed Appendix 2, Procedure 6: Expediting the issuance of a Food Recall Warning to further accelerate the process of issuing a public warning for higher profile situations (for example, incidents involving infant food, where there has been reported illness for E. coli O157:H7 in ground beef or for Listeria monocytogenes in a ready-to-eat food, etc.) so that a public warning can be issued almost immediately after a health risk is confirmed.
In 2015, the CFIA issued the Protocol for CFIA's Sharing of Information during Food Safety Investigations and Recalls. The protocol describes the type of information that can be shared and the type of information that must be protected at various stages of the food safety investigation and recall process, according to guiding principles and legal obligations. It acknowledges the well-defined purpose for communicating with the public during a food recall. It also recognizes that during an active food safety investigation (before a health risk is confirmed), the CFIA's obligation to protect confidential business information and personal information, and the need to protect the integrity of the investigation, significantly limits the ability to release information.
Despite these constraints, there are circumstances where specific food safety incidents may require public communication in the early stages of the food safety investigation, in other words, before the level of health risk has been established and/or a food recall is implemented. These guidelines were developed to address these situations, referred to as early public communication. The guidelines address two types of situations: (1) early public communication to respond to food tampering situations and; (2) early public communication to address concerns raised by the public regarding food safety incidents. The guidelines provide the factors to guide decision makers in determining the need for early public communication, the communication objectives and other considerations for both types of situations.
Guiding principles
The Protocol for CFIA's Sharing of Information during Food Safety Investigations and Recalls outlines the guiding principles as well as CFIA's authorities and responsibilities regarding the sharing of information during food safety investigations and recalls. These guidelines align with the Protocol and provide further details to assist CFIA decision makers and to promote a consistent application.
Roles and responsibilities
Communications and Public Affairs Branch
- Monitors public and media inquiries, and social and traditional media to identify situations where an incident, or alleged incident, is generating public attention; and will liaise with Operations Branch, Office of Food Safety and Recall (OFSR).
- Works with Operations Branch on the appropriate communications approach, including communications products and distribution plans.
Operations Branch
- Leads food safety investigations and identifies emerging incidents where an early public warning may be warranted; informs Communications and Public Affairs Branch of imminent public releases from Canadian authorities or foreign countries; and verifies whether the incident is being investigated by the inspectorate.
- Liaises with other Branches as appropriate.
Policy and Programs Branch
- Provides assistance as required when an early public warning is being considered.
- Liaises with national industry associations as appropriate.
Science Branch
- Provides assistance to Operations Branch and Communications and Public Affairs Branch as appropriate when an early public warning is being considered.
Approvals
- The Vice-President, Operations Branch and the Vice-President, Communications and Public Affairs Branch approve the information released.
1.0 Early public communication associated with food tampering
The CFIA addresses all potentially high health risk incidents on a high priority basis so that any risk mitigation measures can be implemented without delay. In exceptional situations where food tampering is suspected or confirmed, public communication before a health risk is confirmed may be required.
1.1 Decision considerations
Early public communications may be warranted if:
- The CFIA is aware of multiple reports of potential/confirmed food tampering related to a particular food, where the situation may cause significant adverse health effects, or concerns within the population
- Evidence collected through food safety investigation is not sufficient for assessing the health risk, therefore, it is not possible to issue a Food Safety Warning or a Food Recall Warning using Appendix 2, Procedure 6: Expediting the issuance of a food recall warning
- There is a benefit to communicating with the public to provide risk mitigation advice
- Releasing information will not interfere with police investigations
1.2 Communication objectives
- When applicable, inform Canadians of potential risks of injury to human health.
- Demonstrate the Government of Canada's commitment to food safety.
1.3 Communication considerations
- The communication product must not jeopardize or cause interference in an investigation being undertaken by a law enforcement authority.
- Any personal information or confidential business information must not be released unless it is relevant for risk mitigation purposes (consult with CFIA Legal Services, as appropriate).
- When information is circulating in the public domain, messaging will attempt to reassure the public that the CFIA is aware of the incident, is providing assistance to law enforcement authorities (if applicable), and will take appropriate actions as necessary.
- When appropriate, joint communications will be explored with law enforcement authorities and the implicated regulated parties.
- When applicable, messaging will reflect that contained in any public communication issued by the lead law enforcement authority.
- Communications products will provide consumers with the necessary information (based on verified facts) that they need to protect themselves.
- Communications products may include a call to action such as requesting consumers to report related incidents to the CFIA or to regulated parties.
- When possible, relevant regulated parties, industry or consumer associations and, as required, Federal/Provincial/Territorial partners will be advised or consulted before issuing the public communication.
- Opportunities for broadening reach via partners' social media channels will be explored.
1.4 Communications products for consideration
- Consumer Advisory (via web posting, email notification, dissemination to media).
- Using social media (via CFIA channels).
- Posts/tweets to share the CFIA's statement (advisory) and to respond directly to any inquiries
2.0 Early public communication to address concerns raised by the public
Early public communication during a food safety investigation may be required to address (or prevent) concerns raised by the public. Such situations may begin as isolated incidents brought to social or traditional media and may generate significant public attention. The real or potential health risk associated with a food safety incident may be low, but the public may have a different perception. Concerns may also be raised as a result of public announcements in foreign countries or by other Canadian authorities. Such situations may include:
- Consumer complaints or other food safety incidents (in Canada or abroad) that are communicated through social or traditional media and subsequently generate significant attention or
- Public statements/advisories issued by foreign countries or other Canadian authorities which raise, or may raise, concerns regarding the safety of the Canadian food supply
2.1 Decision considerations
The CFIA's initial response to such situations is to respond directly to individual complainants or inquiries received through social or traditional media. Should the situation warrant it, the CFIA's response will gradually escalate. Typically in these situations Communications and Public Affairs Branch develops media lines and key messages to be used in CFIA responses. However, there may be situations where a pro-active approach, using early public communication, is warranted when:
- The food complaints/incidents are generating significant concerns from the public and/or the CFIA is receiving a high volume of inquiries
- The CFIA is aware that a public announcement from a foreign country or a Canadian authority is imminent and the release may generate significant concern from the public or
- Not responding promptly may generate doubt from the public regarding CFIA's ability to respond, or may perpetuate an inaccurate public risk perception
2.2 Communication objectives
- Address Canadians' perception of risk by providing balanced information.
- Inform the Canadian public regarding a food safety incident.
- Demonstrate the Government of Canada's commitment to food safety.
2.3 Communication considerations
- Any personal information or confidential business information must not be released (consult with CFIA Legal Services, as appropriate).
- Communications products will reassure the public that the CFIA is aware of the incident and is taking appropriate actions.
- When necessary, messaging will acknowledge consumers' perception of risk while characterizing the situation in an appropriate manner according to the potential level of health risk.
- When possible, relevant regulated parties, industry or consumer associations and, as required, Federal/Provincial/Territorial partners will be advised or consulted before issuing the public communication.
- Opportunities for broadening reach via partners' social media channels will also be explored.
2.4 Communications products for consideration
- Consumer Advisory (via web posting, email notification, dissemination to media).
- Using social media (via CFIA channels).
- Posts/tweets to respond directly to concerned citizens and to share the CFIA's statement (advisory), if applicable.
- Responses to public inquiries based on messaging in consumer advisories.
Procedure 9: Mandatory recalls
Accessible only on the Government of Canada network.
Procedure 10: Determining after-hours work when responding to food safety incidents
Purpose
To provide the Canadian Food Inspection Agency (CFIA) Operations Branch with guidance for determining which food safety incidents are to be addressed outside of core business hours.
Overview
This procedure provides criteria for identifying high priority food safety incidents and outlines the process to determine when to conduct activities outside of core business hours, including factors to be considered in making the decision.
Background
It is important for management responsible for Operations Branch resources to effectively respond to food safety incidents and allocate resources in a way to achieve food safety objectives. There are a number of stakeholders engaged within Operations Branch to address food safety incidents. Strong collaboration is important between these units in order to provide support as required and ensure a seamless and efficient food safety response.
At all times, high priority food safety incidents will take precedence over other work during core business hours with a view to minimize possible after-hours work. In certain situations, working outside of core business hours may be required in the Area(s) or at the Office of Food Safety and Recall (OFSR) or by both. This guideline facilitates consistent decision making for determining and approving overtime for food safety investigations and recall activities.
Authorities
Overtime approval will be done by management with the delegated authority for the direction of human and financial resources (see the CFIA Authorization of Overtime Policy).
Guideline
Guiding principles
The decision for engaging CFIA Operations personnel to respond to food safety incidents outside of their respective core business hours is guided by the following principles:
- Respond appropriately – the CFIA's response will be proportional to the level of risk posed by the food/hazard combination in question together with other factors that are pertinent to the issue
- Take actions to remove unacceptable or possibly unacceptable hazard exposure for the consumer in a timely manner
- Effective and efficient use of Agency resources to achieve food safety objectives – business requirements should be addressed during core business hours to the extent possible
- The health and well-being of staff working in the Operations Branch is a priority. Occupational health and safety considerations (for example, staff wellness, minimizing excessive work hours) must be integrated into the food safety investigation management and decision making process
Determining after-hours work
There are two main purposes for carrying out after-hours activities:
- To address a situation where the degree of urgency is such that it cannot wait for the next business day; or
- To gain efficiency with regard to upcoming activities by minimizing delays in the resolution of an incident or with a view to reduce the overall overtime cost for Operations Branch.
The following outlines the process for determining if work outside of core business hours (in respective time zones) is required by the Areas or the OFSR.
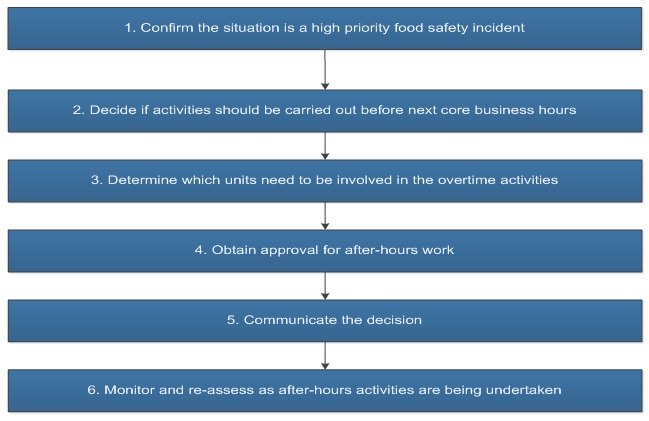
Description of Figure 1: Determining after-hours work when responding to a food safety incident
Figure 1 is a flow chart that outlines the 6 steps involved in determining if after hours work is necessary when responding to a food safety incident. The steps are: confirm the situation is a high priority food safety incident; decide if activities should be carried out before next core business hours; determine which units need to be involved in the overtime activities; obtain approval for after-hours work; communicate the decision; and monitor and re-assess as after-hours activities are being undertaken.
1. Confirm the situation is a high priority food safety incident
The lead investigator, in consultation with their supervisor, will confirm the situation is a high priority food safety incident as listed in Annex A: High priority food safety incidents. Overtime will only be used for high priority food safety incidents. A minimal amount of overtime may be needed to consult and determine whether the incident is a high priority food safety incident.
2. Decide if activities should be carried out before next core business hours
As soon as possible and prior to the end of core business hours the lead investigator and other Operations Branch personnel involved in the file will:
- identify the activities required to advance the food safety investigation (see Annex B)
- define the objective of the activity and the anticipated time of completion with the goal to complete the activity as soon as possible and prior to 10:00 PM local time for employee wellness considerations
- discuss with their respective supervisors/managers (as per Area protocol) to decide if the activities need to be carried out before next core business hours.
The following is used together with the decision tree (see Annex C) to guide the decision by the supervisor/manager.
A. If the presence of a hazard in a food has been confirmed
After-hours activities will be undertaken any day of the week for high priority food safety incidents when the presence of a hazard in a food has been confirmed.
Rationale:
Identified activities are conducted to address a situation where the degree of urgency is such that it cannot wait for the next business day.
Examples of situations:
- A laboratory result confirmed the presence of a pathogen in a food potentially on the market.
- A company initiated a recall due to injurious material in infant food.
- Following the recall of a food sold in bulk or that may be used as an ingredient in other food, conduct food safety investigations at the receiving client level to determine if any secondary recalls are required.
- Following receipt of a foreign notification of a recall for a food that was exported to Canada, conduct verifications to determine if it has been distributed and to take appropriate risk mitigation steps when necessary.
- Following receipt of an unsatisfactory laboratory result on a non-distributed or exported food, conduct preliminary verifications to determine if there is any potentially affected food in distribution that were made under the same conditions (for example, produced on the same line without intervening sanitation, packaged under different brands or formats, etc.).
However, for allergen incidents, Operations Branch will not conduct after-hours activities from Monday to ThursdayFootnote 11 unless:
- the situation is a potential Class I recall where there has been a reported reaction or the food is marketed as "allergen-free"; or
- After-hours work is necessary to meet CFIA performance indicators, such as the issuing of a public warning within 24 hours of a recall determination.
B. If the presence of a hazard in a food has not been confirmed
High priority food safety incidents when the presence of a hazard has not been confirmed may warrant after-hours activities based on the type of incident and the purpose of the after-hours activity.
Rationale:
Identified activities are conducted to either:
- address certain situations where the degree of urgency is such that it cannot wait for the next business day; or
- gain efficiency with regard to upcoming activities by minimizing delays in the resolution of an incident or with a view to reduce the overall overtime cost for Operations Branch.
Situations include:
- Collect information or food samples in relation to a foodborne illness outbreak or confirmed illness
- Sample collection related to a suspected illness associated with an infant food
- Food tampering
- Determining the scope of potentially affected products following a recallFootnote 12 for a high priority incident (may include developing a sampling plan and conducting sampling)
- Exceptional situations
If it is decided that after-hours work is not required at this time then the activities should continue as high priority at the onset of the next core business hours. The need to work after-hours will be re-evaluated the next day.
3. Determine which work units need to be involved in the overtime activities
This determination is made collaboratively by the staff involved in the incident: lead investigator, supervisor, area/regional recall coordinator, OFSR Officer and others as needed. Depending on the activities being performed, after-hours work may not be required by all units involved in a high priority food safety incident. However, when multiple work units are involved, their actions are interdependent and need to be done in a coordinated way.
4. Obtain approval for after-hours work
Before working overtime, each unit involved in the proposed after-hours activities will obtain approval from their responsible manager or director as per their authorities and their respective approval process. The approving manager should ensure that the employees working overtime have the appropriate tools including access to the Government of Canada electronic network if needed.
It is important that all units involved agree on the need to carry out the activities by working overtime in order to achieve a timely and efficient response. If there is disagreement between the Areas, or between the inspectorate and the OFSR with regard to the planned overtime, the issue should be discussed via teleconference by the managers involved. If an agreement cannot be reached, the issue should be brought to the attention of the directors of operations and the director, OFSR as soon as possible to seek resolution.
5. Communicate the decision
When the appropriate approvals have been obtained, the decision to perform the after-hours activities will be communicated by email among the various units involved to facilitate coordination.
6. Monitor and re-assess as activities are being undertaken
The various units involved in the after-hours activities will communicate regularly to inform each other of progress. Reassessment of whether the defined objectives will be met by 10:00 PM local time should be undertaken on an ongoing basis.
After-hours activities will stop under the following circumstances:
- The defined objective has been met;
- Part of the objective was achieved and the next step is awaiting input from external parties (for example, a health risk assessment request was submitted to Health Canada and the response is not expected in a reasonable time frame to complete the defined objective);
- An allergen incident with a reported reaction has been assessed as representing a Health Risk 2 situation unless the next day is not a business dayFootnote 13; or
- The defined objective cannot be accomplished because:
- The implicated regulated party is unavailable
- Critical information or sample results required to complete the set objective within reasonable timelines (before 10:00 PM) are missing
- Specific expertise required is unavailable
If the situation changes, the units involved will discuss when to re-engage. Depending on the day of the week, this could mean continuing that evening, on the weekend or on a statutory holiday.
It is preferred that after-hour activities are completed by 10:00 PM. However, there may be instances where the defined objective can be completed with a minimal amount of additional time (for example, to complete the issuance of a public warning on a Friday evening). In such situations, the involved units will discuss whether overtime continues beyond 10:00 PM in order to minimize the need to deploy resources over the weekend or on a statutory holiday.
In all instances, any unresolved file will be treated as a high priority at the onset of the next core business hours.
Annex A: High priority food safety incidents
A high priority food safety incident is an incident that takes precedence over all other work by Operations Branch.
Type of high priority food safety incidents
- Suspected or confirmed foodborne illness outbreak situation
- Hospitalization or death related to food
- ReportedFootnote 14 illness, injury or reaction related to food including supplemented food
- To help determine if the food is a supplemented food, refer to Supplemented Food Regulations, Appendix 2: Is my product a supplemented food? (accessible only on the Government of Canada network – RDIMDS 17770204)
- See notes below for additional information
- Hazard in food targeted at a vulnerable population, for example:
- the elderly or the immunocompromised
- infants and toddlers
- Tampering incident (suspected or confirmed)
- Company initiated (includes foreign companies) food recall, removal or correction of a marketed food for food safety incidents (all hazards)
- Public warning issued by other government departments (all hazards)
- Food safety incident involving shellfish exported to the United StatesFootnote 15
- Pathogen detected in food or on direct food contact surface (for example, Listeria monocytogenes detected on a slicer)
- Undeclared priority allergen
- Toxin in food (for example, marine biotoxins and Staphylococcus toxins)
- Injurious extraneous material in beverages
- Multiple complaints involving the same food or incident
Notes
- The priority assigned to a food safety incident may change during the course of an investigation.
- In all situations, but particularly complex ones, the lead investigator can consult with the area/regional recall coordinator to determine whether a situation should be treated as high priority. The OFSR is available to assist the area/regional recall coordinator in the determination.
- For more information relating to supplemented foods, please refer to:
- Operational directive: Responding to complaints or enquiries related to supplemented foods amendments to the Food and Drug Regulations during the transition period (accessible only on the Government of Canada network – RDIMS 17594568)
- Amendments to the Food and Drug Regulations related to front-of-package labelling and supplemented foods (accessible only on the Government of Canada network)
Annex B: Food safety investigation activities
Collection of information
Collection of information may be necessary as soon as possible before the risk is confirmed (for example, to confirm the presence of a hazard, to facilitate a health risk assessment request) or after the risk is confirmed (for example, to implement a risk mitigation decision, to verify the extent of the problem).
Planning and guidance
Planning and guidance may be necessary before the next core business hours to carry out the food safety investigation in a concerted or more effective way the following day or next core business hours.
Preparation of sampling plans
Sampling plans may need to be developed as soon as possible (before the next core business hours) to carry out sampling activities without delay and, whenever possible, to minimize having to deploy inspectors for sampling during weekends.
Carrying out sampling activities
Sampling activities may be required as soon as possible in order to confirm the presence of a hazard or because of the nature of the food (for example, perishable). This can include delivery of samples to the laboratory.
Preparation of a health risk assessment request
A health risk assessment request may need to be submitted without delay to determine the level of health risk posed by a food in order to take appropriate risk mitigation actions.
Communication with the regulated party
Immediate communication may be required with the regulated party to request a recall or to assist them in recall implementation activities, including the review of draft recall warnings.
Issuance of recall warnings
Preparation and issuance of public warnings may be required to communicate a risk associated with a food without delay.
Communication with internal and external parties
Immediate communication may be required in order to provide information to senior management, other government departments or to coordinate media responses.
Annex C: Decision tree to determine after-hours work when responding to a food safety incident
Accessible only on the Government of Canada network.
Procedure 11: Referring food safety incidents to the Office of Food Safety and Recall
Purpose
To outline the process for prioritizing food safety incidents (in other words, what is a high priority food safety incident vs. a normal priority food safety incident). Once a food safety incident has been prioritized, this procedure further explains how to refer high priority and normal priority food safety incidents to the OFSR, including which high priority incidents require an early referral to the OFSR.
Scope
This procedure describes the process for the inspectorate to notify the area/regional recall coordinator of food safety incidents and for the area/regional recall coordinator to subsequently refer a file to the OFSR. The procedure also outlines the types of incidents that require an early referral.
There are situations where an incident is brought to the attention of the OFSR first, either by Operations Branch senior management, other CFIA branches, or external partners (such as the Public Health Agency of Canada, foreign governments or international organizations). In such cases, the OFSR engages the inspectorate without delay by communicating the information to the area/regional recall coordinator.
Roles and responsibilities
The OFSR's role when incidents are referred includes:
- Informing internal and external stakeholders where required;
- Providing risk estimation for prioritization;
- Guiding and coordinating food safety investigations;
- Providing guidance and assistance in respect of sampling;
- Determining whether a health risk assessment is necessary and whether the information collected to date is sufficient for a request to Health Canada; and
- Making a recall determination and guiding the lead investigator in the recall implementation.
Procedure
The Process for Referring Food Safety Investigation Incidents to the OFSR is illustrated in Annex B and starts when an incident is triggered. The inspectorate conducts a preliminary assessment which includes assigning one of the following priorities to the food safety incident:
- High Priority with an Early Referral
- High Priority
- Normal Priority
The priority assigned to a food safety incident may change during the course of an investigation.
At any time during a food safety investigation (before or after notification to the area/regional recall coordinator or referral to the OFSR), the inspectorate can seek advice and guidance from the area/regional recall coordinator to determine whether there is a potential health risk and on appropriate next steps. The area/regional recall coordinator can also seek advice and guidance from the OFSR at any time.
A. High Priority with an Early Referral
Please refer to Annex A: High priority food safety incidents with requirements for early referral for a list of types of food safety incidents that require an early referral.
The OFSR's responsibility for coordinating food safety investigations and recalls at the national level includes communication with internal and external partners. Some types of food safety incidents require the Canadian Food Inspection Agency (CFIA) to engage with partners at an early stage. An early referral to the OFSR enables timely engagement with these partners.
A.1 Notify the area/regional recall coordinator
The lead investigator immediately notifies the area/regional recall coordinator, as per the Area's communication protocol, regardless of the amount of information available on the food safety incident. The lead investigator provides the following information:
- Area of concern;
- Trigger;
- Summary of information available to date;
- If illness, injury or reaction has been reported, the number of cases under investigation and the cases' health status, if known;
- Issues Management System (IMS) number, if available; and
- Brief summary of actions taken to date together with the next actions to be taken
The area/regional recall coordinator reviews the information received in the notification. Where necessary, the area/regional recall coordinator requests additional data or information from the lead investigator. If the incident does not require an early referral, the area/regional recall coordinator informs the lead investigator.
A.2 Early referral to the OFSR
The area/regional recall coordinator immediately refers the incident to the national manager, FSIR by telephone, followed by an e-mail flagged as "urgent" to CFIA.OFSR-BSRA.ACIA@inspection.gc.ca. After regular OFSR working hours (after 5:00 PM ET), the referral should be done by telephone using the emergency number (613-720-5087) followed by an email. The subject line of the e-mail should state "Early Referral". The email should include the information that was provided by the lead investigator in step A.1, including actions taken to date and proposed next steps.
Once an incident is referred to the OFSR, a Food Safety Recall Specialist is assigned to the file.
B. High priority
Please refer to Procedure 15, Annex A: High priority food safety incidents for a list of high priority food safety incidents.
B.1 a) Determine if affected food is potentially in distribution
The lead investigator, in consultation with their supervisor, evaluates food safety investigation findings as they are being collected. The lead investigator notifies the area/regional recall coordinator as soon as the information indicates that the affected food is in distribution or potentially in distribution. Food is considered to be in distribution when it has left the direct control of the regulated party responsible for that food.
When it is not possible for the lead investigator to confirm whether the affected food is under control or on hold by the regulated party, the lead investigator will consider the food to be distributed.
If the affected food is in distribution or is potentially in distribution, the lead investigator proceeds to B.2 Notify the area/regional recall coordinator
If the affected food is confirmed to be not in distribution, the lead investigator proceeds to B.1 b) Determine if other affected foods are potentially in distribution
B.1 b) Determine if other affected foods are potentially in distribution
The lead investigator, in consultation with their supervisor, evaluates food safety investigation findings and notifies the area/regional recall coordinator as soon as:
- the information indicates that other affected food has been distributed or could have been distributed; and/or
- the affected food was imported and there could be other shipments distributed by the same importer or other shipments distributed by other importers that could be affected by the same problem.
The lead investigator will notify the area/regional recall coordinator before taking any samples.
If it is determined that no other affected food could have been distributed, the lead investigator follows the established procedures to verify compliance and take appropriate compliance measures.
B.2 Notify the area/regional recall coordinator
The lead investigator immediately notifies the area/regional recall coordinator as per the Area's communication protocol and provides the following information:
- Area of concern
- Trigger
- A summary of the food safety incident and their findings to date
- IMS number
- Brief summary of actions taken together with the next actions to be taken
Once the area/regional recall coordinator is notified of an incident, the area/regional recall coordinator becomes engaged and provides guidance to the inspectorate, as required.
B.3 Refer to the OFSR
The area/regional recall coordinator immediately refers the incident to the OFSR by sending an e-mail flagged as "urgent" to CFIA.OFSR-BSRA.ACIA@inspection.gc.ca. After regular OFSR working hours (after 5:00 pm ET), the referral should be done by telephone using the emergency number (613-720-5087), followed by an email. The subject line of the e-mail should state "High Priority Referral". The email should include the information that was provided by the lead investigator in B.2, including actions taken to date and proposed next steps.
Once an incident is referred to the OFSR, a Food Safety Recall Specialist is assigned to the file. At this stage, the lead investigator and the area/regional recall coordinator may not have all the information required for the OFSR to initiate its activities. The lead investigator will continue to lead the food safety investigation and the area/regional recall coordinator will continue to provide guidance and advice as necessary. The OFSR will provide guidance and will initiate its activities when required.
C. Normal priority
C.1 a) Determine if affected food is potentially in distribution
The lead investigator, in consultation with their supervisor, evaluates food safety investigation findings as they are being collected and notifies the area/regional recall coordinator as soon as the information indicates that the affected food is in distribution or potentially in distribution. Food is considered to be in distribution when it has left the direct control of the regulated party responsible for that food.
When it is not possible for the lead investigator to confirm whether the affected food is under control or on hold by the regulated party, the lead investigator will consider the food to be distributed.
If the affected food is in distribution or is potentially in distribution, the lead investigator proceeds to C.2 Notify the area/regional recall coordinator.
If the affected food is confirmed to be not in distribution, the lead investigator proceeds to C.1.b) Determine if other affected foods are potentially in distribution.
C.1 b) Determine if other affected foods are potentially in distribution
The lead investigator, in consultation with their supervisor, evaluates food safety investigation findings and notifies the area/regional recall coordinator as soon as:
- the information indicates that other affected food has been distributed or could have been distributed; and/or
- the affected food was imported and there could be other shipments distributed by the same importer or other shipments distributed by other importers that could be affected by the same problem.
The lead investigator will notify the area/regional recall coordinator before taking any samples.
If it is determined that no other affected food could have been distributed, the lead investigator follows the established procedures to verify compliance and take appropriate compliance measures.
C.2 Notify the area/regional recall coordinator
The lead investigator immediately notifies the area/regional recall coordinator as per the Area's communication protocol and provides the following information:
- Area of concern;
- Trigger;
- A summary of the food safety incident and their findings to date;
- IMS number; and
- Brief summary of actions taken together with the next actions to be taken.
Once the area/regional recall coordinator is notified of an incident, the area/regional recall coordinator becomes engaged and provides guidance to the inspectorate.
C.3 Determine when to refer to the OFSR
The area/regional recall coordinator evaluates the information received in the notification, and determines when to refer the file to the OFSR.
The file should be referred when:
- Advice or coordination from the OFSR is required to advance the food safety investigation
- Sampling advice or sampling coordination from the OFSR is required
- Hazard is in the process of being confirmed (for example, laboratory results are pending) and product core information (for example, food details and distribution) is available
- Presence of a hazard has been confirmed in a food in distribution. For example, through:
- Unsatisfactory laboratory results
- Inspection observations, such as insect infestation, extraneous material
- Document review, such as calculations of levels of allergens based on formulations and known percentage of ingredient
If it is determined that the file is not ready to be referred, the area/regional recall coordinator reevaluates the file on an ongoing basis. The area/regional recall coordinator may determine that the file does not need to be referred to the OFSR as no hazard was identified in food in distribution or potentially in distribution.
C.4 Refer to the OFSR
The area/regional recall coordinator immediately refers the incident to the OFSR by sending an e-mail to CFIA.OFSR-BSRA.ACIA@inspection.gc.ca. After regular OFSR working hours (after 5:00 pm ET), the referral should be done by telephone using the emergency number (613-720-5087), followed by an email. The subject line of the e-mail should state "Referral". The email should include the information that was provided by the inspector in C.2, including actions taken to date and proposed next steps.
Once an incident is referred to the OFSR, a Food Safety Recall Specialist is assigned to the file.
Annex A: High priority food safety incidents and requirements for early referral
A high priority food safety incident requires immediate and sustained attention. Work on these incidents takes precedence over all other work and may continue after-hours where necessary.
Inspectors should review the entire table below to determine if their food safety incident fits any of the high priority food incidents listed. If it is determined that the incident is a high priority incident, it should be referred to the area/regional recall coordinator for subsequent referral to OFSR however, the timing of the referral is dependent on if an early referral is required or not, as outlined by the second column.
Notifications for high priority incidents that require an early referral will be made to the area/regional recall coordinator immediately, regardless of the amount of information available on the food safety incident. The area/regional recall coordinator will refer the incident immediately to the OFSR to initiate collaboration and gain efficiency in the resolution of the incident.
Notifications for high priority incidents that do not require an early referral as well as normal priority incidents will be made to the area/regional recall coordinator as soon as it is determined that affected food has been or could have been distributed. The area/regional recall coordinator will refer high priority incidents immediately to the OFSR, and will refer normal priority incidents after evaluating the information.
| Type of high priority food safety incidents | Early referral required? |
|---|---|
| Suspected or confirmed foodborne illness outbreak situation | Yes |
| Hospitalization or death related to food | Yes |
Reported Table Note c illness, injury or reaction related to food including supplemented food
|
Yes |
Hazard in food targeted at a vulnerable population, for example:
|
Yes |
| Tampering incident (suspected or confirmed) | Yes |
| Company initiated (includes foreign companies) food recall, removal or correction of a marketed food for food safety incidents (all hazards) | Yes |
| Public warning issued by other government departments (all hazards) | Yes |
| Food safety incident involving shellfish exported to the United States Table Note d | Yes |
| Pathogen detected in food or on direct food contact surface (for example, Listeria monocytogenes detected on a slicer) | No |
| Undeclared priority allergen | No |
| Toxin in food (for example, marine biotoxins and Staphylococcus toxins) | No |
| Injurious extraneous material in beverages | No |
| Multiple complaints involving the same food or incident | No |
Notes
- The priority assigned to a food safety incident may change during the course of a food safety investigation.
- In all situations, but particularly complex ones, the lead investigator can consult with the area/regional recall coordinator to determine whether a situation should be treated as high priority. The OFSR is available to assist the area/regional recall coordinator in the determination.
- For more information relating to supplemented foods, please refer to:
- Operational directive: Responding to complaints or enquiries related to supplemented foods amendments to the Food and Drug Regulations during the transition period (accessible only on the Government of Canada network – RDIMS 17594568)
- Amendments to the Food and Drug Regulations related to front-of-package labelling and supplemented foods (accessible only on the Government of Canada network)
Annex B: Process for referring food safety incidents to the OFSR
Accessible only on the Government of Canada network.
Appendix 3: Templates / Forms
Template 1: Food safety investigation plan recording form
Accessible only on the Government of Canada network.
Template 2: Letter template for a confirmation of a recall
Accessible only on the Government of Canada network.
Template 3: Letter template for a company initiated recall
Accessible only on the Government of Canada network.
Template 4: Notice of recall
Urgent – Recall of (enter food name)
(Enter date)
(Enter name and address of your business)
Dear Customer or Attention (enter name of customer contact),
(Enter your business name) is recalling the food listed below because (state the reason for the recall, for example, presence of an allergen not declared on the label, bacteria [name], foreign pieces of material, etc.)
| Brand name | Common name | Size | Lot code(s) on food | UPC |
|---|---|---|---|---|
Please discontinue the distribution, sale or use of this food immediately. Remove food from stock or display, count the quantity in your inventory and ensure that the food will no longer be distributed, sold or used. Please do this by [insert one of the following two sentences]
[1] storing all recalled food in a secure, segregated place and mark as "recalled"
[Include a paragraph providing information, as appropriate, regarding your business' policy on crediting for recalled food, identification of recalled food with the mark "recalled" and arrangement for the pick-up recalled food. Example: We will credit you for the recalled food. Please mark the food "recalled" and our staff will call you to arrange pick up.]
[2] proceeding with the destruction of all recalled food in such a manner that they cannot be sold or used.
[Include a paragraph providing information, as appropriate regarding your business' policy on crediting for recalled food]
If you further distribute this food, please immediately contact all your clients to whom you distributed this food and inform them of this recall and actions they should take on the food.
If any of the recalled food is repackaged, relabelled or reworked by your business, your final product may also pose a health risk. Please contact your local CFIA office immediately should this situation apply.
Important
Please acknowledge receipt by signing and sending this document (via fax or e-mail) to (enter your business' contact name) at (enter your business' fax number or e-mail address).
Date / Time Received: __________
Name (please print): __________
Business name: __________
Signature: __________
Thank you for your cooperation.
[Sign here]
(Enter your business' contact, their position and your company name)
Template 5: Letter template to distributors requesting recall action
Accessible only on the Government of Canada network.
Template 6: Letter template for a confirmation of a market withdrawal
Accessible only on the Government of Canada network.
Appendix 4: Tables
Table 1: Summary of recall options for health risk 3 situations
Accessible only on the Government of Canada network.
Table 2: Summary of product action communication activities
Accessible only on the Government of Canada network.
- Date modified: